Reactive Solutions - An FHWA Technical Update on Alkali-Silica Reactivity
Summer 2008 - Volume 1, Issue 3
Inside this Issue:
- What is ASR?
- What Does ASR Look Like?
- Editor's Corner
- Ask The Experts
- How Do You Diagnose ASR?
- An Update on FHWA's ASR Development and Deployment Program
- This issues's Ask.Send.Receive.
- Schedule of Events
- This Issue's Photo
What is ASR?
Alkali-silica reactivity (ASR) is a type of distress causing premature deterioration that affects numerous concrete structures around the world. Structures affected with ASR will begin to exhibit cracking, and in heavily distressed structures, movement due to excessive expansion.
How exactly does ASR occur? It is well accepted that the three components necessary to induce ASR damage in concrete are:
- Reactive silica (from aggregates)
- Alkalies (mainly from portland cement)
- Sufficient moisture
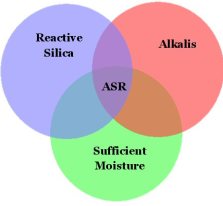
Deleterious aggregates will break down when exposed to highly-alkaline pore solution in concrete and react with sodium and potassium to form the ASR gel. Alkalis are mostly contributed by the portland cement, yet other alkali sources may include chemical admixtures, supplementary cementing materials, external sources (such as deicing salts and seawater), aggregates, and wash water. When considering alkali levels, it is important to look at the total alkali content of the concrete mixture and not solely at the alkali content of the portland cement. In order for ASR to cause damage in concrete structures, there must be available moisture.
Typically, concrete mixtures with high-alkali cements and reactive aggregates show minimal or no expansion when placed in very dry environments. There are cases where part of a structure is exposed to a continual source of moisture (for example due to poor drainage); the section exposed to moisture will exhibit ASR-damage while the rest of the structure will show little or no damage.
Expansion of the gel occurs due to the imbibing of available water. Research has shown that a relative humidity in the concrete above 80% would be sufficient to cause significant expansion due to ASR.
Unfortunately, ASR cannot be repaired - but it can be managed, such as minimizing the availability of moisture, applying coatings such as silane, and confining the expansion. In addition, certain precautions can be taken to prevent ASR in new concrete, such as using supplementary cementing materials, non-reactive aggregates, limiting the total alkali content of the concrete, and using lithium compounds. The next pages discuss what ASR looks like and ways to properly diagnose whether the distress you see is indeed ASR.
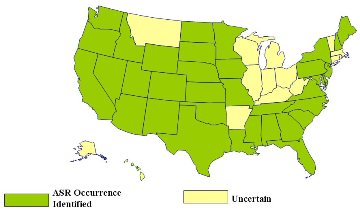
Occurrences of ASR in the United States (from 1994 FHWA Showcase Workshop on ASR)
What Does ASR Look Like?

Figure 1. Core from bridge deck showing vertical surface cracks induced by general expansion and cracking in the interior.
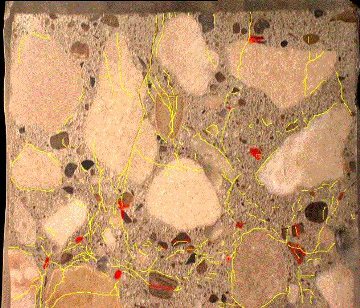
Figure 2. Polished slab of concrete showing cracks through aggregate particles and paste (stained yellow) and alkali-silica reaction gel (stained red).
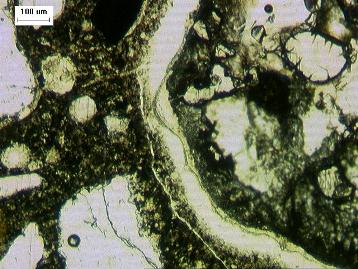
Figure 3. Thin section of concrete showing crack lined with alkali-silica reaction gel.

Figure 4. Polished slab from concrete pavement showing expansive cracking of coarse aggregate particles.

Figure 5. Lapped section from core of damaged bridge deck treated with sodium cobaltnitrite.

Figure 6. Close-up of Figure 5 showing positive indication for potassium in void-filling deposit, along crack in aggregate and in paste around aggregate.
Editor's Corner
Dear Readers,
Welcome to the third issue of Reactive Solutions! This issue is being released just weeks after the 13th International Conference on Alkali-Aggregate Reactions (ICAAR) held in beautiful and historic Trondheim, Norway. The conference was attended by 175 delegates from 28 countries including the U.S.A. and Canada. The conference proceedings, which are available on CD by contacting the editor-in-chief at maarten.broekmans@ngu.no, contain 133 papers reporting the latest findings on alkali-aggregate reaction. It was certainly pleasing to see that a number of North American scientists and engineers are at the forefront of ASR research and that the University of Texas in Austin has been selected as the venue for the 14th ICAAR in 2012. Hope to see y'all at that one!

Dr. Michael Thomas, University of New Brunswick.
Ask The Experts
"I have heard that a compound called lithium can stop ASR. Is this true and how does it work?"
Submitted by Michael Hunt, Permanently Paved, Inc.
Yes, when used appropriately certain lithium compounds can prevent deleterious expansion of concrete containing some types of reactive aggregate. The ability of lithium to control ASR expansion was first reported in 1951 by two researchers, McCoy and Caldwell, with the Lehigh Portland Cement Company in Copley, Pa. The benefits of lithium have been confirmed by many workers since then and lithium-based admixtures for controlling expansion due to ASR are now commercially available in North America.
Lithium is an alkali metal just like the sodium and potassium found in Portland cement which participate in alkali-silica reactions in concrete. However, unlike sodium and potassium, it is thought that lithium forms an insoluble and non-swelling lithium silicate when it reacts with alkali-silica reactive aggregates in concrete and that this product forms a protective barrier around the remaining reactive silica preventing deleterious reaction with the sodium and potassium hydroxides. This is one of the mechanisms currently favoured to explain the role of lithium, but there are others. Unfortunately, lithium does not appear to be equally effective with all types of alkali-silica reactive aggregates and testing is required to determine the suitability of lithium and the dosage required with each source of reactive aggregate.
Got a question? Finding it difficult to get a clear-cut answer?
This section of the newsletter is dedicated to answering your ASR-related questions. In each issue, our editorial committee selects and answers one question submitted by you, the reader. Ask one of our experts-we're here to listen.
Email your question to asrnewsletter@transtec.us.
How Do You Diagnose ASR?
D. Stephen Lane, Virginia Transportation Research Council
When a concrete pavement or structural element exhibits map or pattern cracking, either random or with preferential orientation, it is important to determine the actual causes of the distress. The cracking of the outer surfaces of the concrete tell you that it has undergone differential volume changes between the outer skin and the interior mass, with the "skin" shrinking relative to the interior. However, it doesn't tell you whether in fact the skin shrank or the interior swelled, nor does it tell you what caused the differential volume change. Figure 1 (from article What Does ASR Look Like?) shows a core where internal expansion and cracking has resulted in surface cracking of a concrete bridge deck.
To determine the cause of the cracking, one must examine cores for petrographic evidence of the mechanisms involved. Examples of typical distress manifested by ASR are shown in Figures 2-3 (What Does ASR Look Like?), Handbook for the identification of alkali-silica reactivity in highway structures (SHRP-C-315) and State-of-the-Art report on alkali-aggregate reactivity (ACI 221.1R).
Guidance on procedures and practices to follow in determining the cause of pavement distress can be found in Guidelines for Detection, Analysis, and Treatment of Materials-related Distress in Concrete Pavements, Volumes 1 and 2 (FHWA-RD-01-163, 164). Information on equipment, procedures and practices in performing petrographic examinations of concrete are available in Petrographic Methods of Examining Hardened Concrete: A Petrographic Manual (FHWA HRT-04-150).
In order to determine that the distressed concrete has been affected by ASR, there must be evidence indicating that the damage was caused by aggregate particles reacting to form the ASR gel. Evidence of affected aggregate particles includes cracking, inter-granular corrosion, and sometimes development of rims, particularly on crushed aggregate particles. This type of evidence should also go hand-in-hand with cracking that extends through the paste to indicate that the ASR has resulted in damage to the concrete itself. This type of cracking is best viewed on lapped slabs (Figure 2 in What Does ASR Look Like?) and thin sections (Figure 3 in What Does ASR Look Like?). Other deterioration mechanisms that cause damage by swelling of the paste alone will exhibit cracks that extend through the paste; however they will bypass the aggregate particles resulting in peripheral gaps around the particles.
Identifying ASR constituents in the aggregate and finding reaction gel in the concrete provides the distinction between ASR and other aggregate-related deterioration mechanisms, such as alkali-carbonate reaction and aggregate unsoundness (which also typically exhibits a distinctly different outward manifestation referred to as D-cracking) (see SHRP-C-315).
Chemical spot tests (stains) can be useful in locating reaction product, especially if it is sparse. The spot tests work on the basis of ion exchange and thus provide an indication of the accumulation of the particular ion in the material at a specific location. Two stains are useful in diagnosing ASR: sodium cobaltinitrite, specific for potassium (K); and uranyl acetate, specific for sodium (Na). Of these, the sodium cobaltinitrite is more user friendly since it does not have the environmental issues of uranyl acetate and it imparts a yellow stain visible in ordinary light to K-bearing materials (Figures 5 and 6 in What Does ASR Look Like?) whereas a UV light is needed to examine specimens treated with uranyl acetate. Additionally, potassium is the more prevalent alkali metal in North American portland cements, making it the logical choice to focus on. Keep in mind that the spot test merely indicates the presence of the specific ion in the stained material and that further analysis is necessary to confirm that its characteristics are consistent with ASR gel.
A.S.R. - Ask.Send.Receive.
This Issue's Question

"Is there one specific test that your state uses that lets you know you have ASR?"
Submit your answers to asrnewsletter@transtec.us.
An Update on Federal Highway's ASR Development and Deployment Program
ASR Reference Center
State engineers and practitioners are in need of resources and information related to ASR that is easily accessible. FHWA is responding by developing an ASR Reference Center. Through FHWA's ASR website you will be able to access a broad range of information related to ASR. Some of the resources that will be included in this Reference Center are:
- Research reports related to ASR
- List of reference documents
- List of local, national, and international specifications
- Links to other ASR related websites
- Summaries of past field trials for ASR mitigation
The FHWA wants to know what would be most useful to you. Please provide suggestions on what you would like to see in the Reference Center to Gina Ahlstrom at gina.ahlstrom@dot.gov.
For more information about the progress of this program, visit: https://www.fhwa.dot.gov/pavement/concrete/asr.cfm or contact Gina Ahlstrom at gina.ahlstrom@dot.gov.
Schedule of Events
July
20-23 6th International Conference on Road an Airfield Pavement Technology, Saporro, Japan
August
3-8 AASHTO Subcommittee Meeting on Materials, Asheville, NC
13-15 2nd Workshop on Advanced Characterization, Modeling, and Design of Concrete Pavements, Richmond, California
17-20 Institute of Transportation Engineers 2008 Annual Meeting and Exhibit, Anaheim, California
September
30-October 2 8th International Conference on Creep, Shrinkage, and Durability of Concrete and Concrete Structures, Ise-Shima, Japan
Feature Photo Section
Have a picture you'd like to send us of an ASR-affected structure in your state? Send us what you like - a bridge, a core, a snapshot of your lab, etc. - our editorial committee will select one picture and post it here for all to see.
Email your pictures at asrnewsletter@transtec.us.

Wall built with ASR susceptible aggregate showing ASR gel extruding from the cracks. (Photo taken during the 13th ICAAR field trial trip in Norway, submitted by Gina Ahlstrom.)
Editor: Dr. Michael Thomas (University of New Brunswick, mdat@unb.ca)
Editorial Committee: Gina Ahlstrom (FHWA), Lizanne Davis (FMC Corporation), Steve Lane (Virginia DOT), Colin Lobo (National Ready Mix Concrete Association), Brian Merrill (TxDOT), Peter Taylor (CP Tech Center), Paul Tennis (Portland Cement Association), Leif Wathne (American Concrete Pavement Association).
Contact Information
Gina Ahlstrom, Concrete Pavement Engineer (Gina.Ahlstrom@dot.gov.) FHWA-Office of Pavement Technology
Get On the Reactive Solutions Mailing List
Want to subscribe to future issues? Email us at asrnewsletter@transtec.us. - Subscriptions are free.

