Reactive Solutions - An FHWA Technical Update on Alkali-Silica Reactivity
Volume 3, Issue 2
Inside this Issue:
- Texas DOT Using More Rapid Method to Locate Alkali-Silica Gel
- Editor's Corner
- New International Alkali Aggregate Reaction Center Established
- Ask The Experts
- This Issue's Ask.Send.Receive
- Ongoing FHWA Research
- Schedule of Events
- This Issue's Photos
Texas DOT Using More Rapid Method to Locate Alkali-Silica Gel
New technique quick, simple and effective
The Texas Department of Transportation (TxDOT) has established a new method for identifying Alkali-Silica reactivity (ASR) in the early stages of development. This new method allows for easier identification of the specific reactive particles by making the ASR gel in the concrete sample more visible.
This technique consists of simply placing a polished concrete sample suspected of having ASR problems in a fog room prior to the petrographic analysis. Figure 1 contains photographs of a reactive particle exhibiting ASR before and after placement in the fog room. Prior to this discovery, the petrographer had difficulty isolating the cause of cracking since the amount of gel visible was limited and only associated with air voids; there was little direct evidence that the aggregate was experiencing dissolution. However, when the specimen was examined after being placed in the fog room, the gel was more visible, allowing an easier identification of the source of the gel. The duration of the specimen in the fog room is dependent upon the sample and the extent of ASR. The sample is generally monitored on a daily basis and will begin evaluation once ASR gel is observed, which may be as short as one day to as long as a week.
Figure 1. Comparison between sample before placement in the fog room (left) and after placement in the fog room (right).
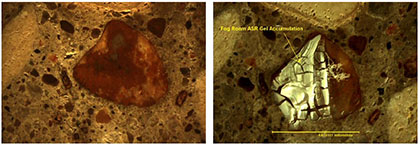
Before the chemistry of the gel observed is confirmed with scanning electron microscope (SEM) or energy dispersive spectrometer (EDS), the samples are dried in an oven at a low temperature. The only discovered drawback of the method is when the sample is left in the fog room too long, resulting in copious amounts of gel observed. In such cases, it is difficult to determine which aggregate is responsible for generating the gel. Figure 2 on the next page illustrates the abundant amounts of gel that can be observed. This method is useful because the early identification of ASR gel in concrete can be difficult to determine properly, even when using petrographic techniques.
Figure 2. Visible ASR gel after placement in the fog room.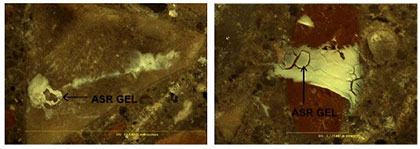

Typical aggregate distress features often appear to be non-existent in the early ages of ASR, such as cracking within the aggregate, propagation of cracking into the paste, and exudation of gel from the reactive particle. When gel is observed in cases such as these, it is usually confined to accumulation in the air voids; as a result, this does not provide conclusive evidence that the concrete distress is caused by ASR gel expansion. In addition, preparation of a sample for petrographic analysis may tend to remove any small quantities of gel and induce micro-cracking, resulting in uncertainty as to whether sample preparation or ASR is the cause of the cracking. Moreover, it is difficult to determine which aggregate particles may be contributing to ASR, making it difficult to identify ASR in early or mild cases under typical thin section work or SEM/EDS analysis.
The new technique is a quick, simple, and effective way to allow for a definitive identification of which particles are producing ASR gel. It also assists in accurately identifying the quantity of potentially reactive products, and confirms presence of ASR in very early stages of development. Staff from TxDOT's Concrete Laboratory and Petrography Laboratory will continue to refine any and all of their techniques used to predict, prevent, and identify ASR distresses in concrete. For more information, contact the Concrete Materials branch of the Construction Division's Materials and Pavements Section at (512) 506-5856.
Editor's Corner
Dear Readers,
March was a busy month for alkali-aggregate reactivity (AAR). ASTM launched a new Subcommittee C09.50 on Risk Management for Alkali Aggregate Reactions, chaired by Steven Parker of CHRYSO Inc., to develop standards on the mitigation of deleterious alkali-aggregate reactions in concrete. A workshop on Specifications on Alkali-Aggregate Reaction will be held at the ASTM meeting in St Louis this June to mark the launch of the new subcommittee. We wish Steve the best of luck in this much-needed endeavor! More information on the subcommittee and workshop can be found at www.astm.org/COMMIT/C09. Also, this March the editor was lucky enough to visit beautiful Azores to attend a meeting and workshop of the RILEM Technical Committee 219—ACS on Alkali Aggregate Reaction in Concrete Structures: Performance Testing and Appraisal. Great strides are being made in Europe regarding testing and preventive measures for AAR and in the evaluation and modeling of structures affected by AAR. Six people from North America attended the meeting and participated in the workshop to ensure that the excellent technical exchange between Europe and North America continues.
Sincerely,
Mike Thomas
Dr. Michael Thomas, University of New Brunswick.

New International Alkali Aggregate Reaction Center Established
The International Centre of Research and Applied Technology for Alkali Aggregate Reactions (AAR) has been established in Iceland at the laboratory of Mannvit Engineering.
The Centre, founded by Professor Børge Johannes Wigum, was established to implement decades of domestic and international research through individuals and organizations. The Centre partners with various international experts in AAR, representing research organizations and companies, as well as universities, in North America, Europe, and Asia. Laboratory staff is composed of several skilled experts in the fields of concrete, aggregates, geological and geotechnical engineering.
For more information about the Centre, please visit http://www.mannvit.com/TestingResearchLab/CentreofResearchandTechnologyforAAR/or contact Dr. Børge Johannes Wigum at wigum@mannvit.is.
Ask The Experts
Do deicers or salts used during winter maintenance have any effect on ASR?
This is still a source of debate amongst researchers. A number of studies/papers suggest that exposure to de-icing salt can initiate or accelerate deterioration in concrete due to ASR by supplying additional alkalies. This would occur when the application of de-icing salt contributes in increasing the availability of OH— ions (or pH) in the pore solution of the concrete. Using sodium chloride as an example, for this to happen in concrete, however, the Cl— ions from the salt need to be bound by cementitious phases (e.g. forming Friedel's salt, i.e. calcium—chloroaluminate phase), thereby leaving a surplus of Na+ ions in solution, which can be balanced by negatively charges ions, such as OH—. The extent to which NaCl will increase ASR damage is thus largely dependent on the amount of salt solution that can infiltrate the concrete and the amount of C3A present in the cement. In the process of salt application on concrete incorporating a reactive aggregate, the expansion (and cracking) of the concrete can also be partially caused by the formation of expansive chloroaluminate phases in the hardened concrete.
Overall, the effect of NaCl on expansion due to ASR is affected by a number of factors, such as the composition of the cement (C3A and alkali content), the type and nature of reactive aggregate (some aggregates seem to be more sensitive than others), the concrete alkali content, the time of addition or exposure of the concrete member to salt, and the permeability of the concrete. The complex nature of this interaction likely explains the somewhat contradictory information reported in the literature on the topic.
Got a question? Finding it difficult to get a clear-cut answer?
This section of the newsletter is dedicated to answering your ASR-related questions. In each issue, our editorial committee selects and answers one question submitted by you, the reader. Ask one of our experts-we're here to listen.
Email your question to asrnewsletter@transtec.us
A.S.R. — Ask.Send.Receive.
This Issue's Question
Does your state use any established testing methods for job mixes to test for ASR?
Submit your answers to asrnewsletter@transtec.us

Ongoing FHWA Research
New technology used to rehabilitate ASR-affected pavement
The Federal Highway Administration's ASR research program is currently conducting research aimed at providing a more rapid assessment of the alkali-reactivity of aggregates using non-destructive evaluation (NDE) methods. Research performed by the Georgia Institute of Technology has already demonstrated that NDE methods based on non-linear acoustics are able to accelerate aggregate screening for Alkali-Silica reactivity (ASR) as tested by the American Standards for Testing and Materials (ASTM) C1260 test method. In as early as six days, aggregates with varying reactivity were discriminated through the use of an NDE-defined ASR damage index, suggesting that using acoustic methods alongside the standard accelerated mortar bar test could cut the testing period in half.
The ongoing FHWA research focuses on utilizing NDE methods to more rapidly determine the reactivity of aggregates tested by ASTM C1293 (concrete prism test). This research indicates that ASR damage in concrete (as a measure of expansion) directly relates to changes in resonance frequency with increasing amplitude of excitation. As a result, this method can be used as an index to characterize the damage in concrete specimens.
Non-linear acoustic measurements are also being conducted on concrete samples cast with fine and coarse aggregates of varying reactivity. The goal of this testing is to better understand the relationships between aggregate reactivity and the measured changes in non-linearity. This work would ultimately lead toward the development of a non-destructive acoustic measurement, which could then be used in conjunction with standard concrete test methods to provide an earlier indication of aggregate reactivity.
For more information about the status of this research, please contact Prof. Kimberly Kurtis with the Georgia Institute of Technology at kimberly.kurtis@ce.gatech.edu.
Schedule of Events
May
5-7 TRB First International Conference in North America on Nanotechnology in Cement and Concrete, Irvine, California
9-12 International Symposium on Cement and Concrete, Jinan, China
June
6 ASTM Workshop on Specifications on Alkali—Aggregate Reaction, St. Louis, Missouri
16-18 ACPA Mid—Year Meeting, Meeting Site to be Announced
July
11-14 WASHTO Annual Meeting, Bismark, North Dakota
11-15 Fifth International Conference on Bridge Maintenance, Safety, and Management, Philadelphia, Pennsylvania
This Issue's Photo
Figure 3. Concrete foundation of a storage tank in Hunt, Virginia constructed in 1993.

Figure 4. Close-up view of ASR-distressed concrete. Coarse aggregate is a granitic gneiss.

Both photos sent by Nick Scaglione, Concrete Research and Testing, LLC.
Have a picture you'd like to send us of an ASR-affected structure in your state? Send us what you like — a bridge, a core, a snapshot of your lab, etc. — our editorial committee will select one picture and post it here for all to see. Email your pictures to asrnewsletter@transtec.us.
Technical Editor: Dr. Michael Thomas
Managing Editor: Bebe Resendez
FHWA Contact: Gina Ahlstrom, Concrete Pavement Engineer (Gina.Ahlstrom@dot.gov.) FHWA-Office of Pavement Technology
Editorial Committee: Lizanne Davis (FMC Corporation), Steve Lane (Virginia DOT), Colin Lobo (National Ready Mix Concrete Association), Brian Merrill (Texas DOT), Peter Taylor (CP Tech Center), Paul Tennis (Portland Cement Association), Leif Wathne (American Concrete Pavement Association)
Get On the Reactive Solutions Mailing List
Want to subscribe to future issues? Email us at asrnewsletter@transtec.us. - Subscriptions are free.

