U.S. Department of Transportation
Federal Highway Administration
1200 New Jersey Avenue, SE
Washington, DC 20590
202-366-4000
Federal Highway Administration Research and Technology
Coordinating, Developing, and Delivering Highway Transportation Innovations
 |
| This report is an archived publication and may contain dated technical, contact, and link information |
|
Publication Number: FHWA-RD-97-146 Date: NOVEMBER 1997 |
Previous | Table of Contents | Next
The concrete petrographer is often called upon to ascertain the presence or absence of particulate materials other than portland cement in a specimen of HCC. These materials include all the finely comminuted solid substances that may be added to HCC to improve its properties or reduce its cost. These substances are of relatively small grain size, usually at least as small as that of portland cement. Included are pulverized limestone (Kleiger & Hooton, 1988), hydrated lime, natural pozzolans, fly ash, GGBFS (Hwang & Shen, 1991), and silica fume (see Tuthill, 1978b; Transportation Research Board, 1990).
In general, when these materials have been used in a concrete mixture as prescribed, the resulting concrete is tougher and less permeable than a similar concrete that does not include these materials. Historically, some of these substances were considered to be adulterants and the use of them was considered an effort to dilute the cement and produce a less costly, less energy-intensive product of unknown quality. However, in general, these materials improve HCC and the major drawback to their use is delayed strength gain (except with silica fume). In cold weather, this delay can be sufficient to mandate against their use. When HCC containing these materials has not attained the expected maturity and is used to support further placements, equipment, or personnel, the results can be disastrous. The petrographer should be aware of these problems. Where young HCC failed to bear the expected load, the cause could be the use of materials whose slow strength gain was not properly allowed for.
These materials should not be considered as adulterants when they have been specified for use and necessary allowances made. The particulate materials that are rich in amorphous silica (GGBFS, fly ash, silica fume, and natural pozzolan) are pozzolanic and will combine with the calcium hydroxide of HCC to form silicate hydrates that are cementitious and indistinguishable from the hydrated products of portland cement. According to B. Mather (personal communication, October 1991), GGBFS is a slowly hydrating hydraulic cement and is, therefore, not a pozzolan, although its action is, in many ways, similar to that of a pozzolan (ACI 226.1R). Such a reaction can, during the early stages of the setting of the cement paste, cause the sequestration of deleterious alkalies in nonswelling silica gels (see Appendix F). These gels may be thinly distributed throughout the paste. Thus, these pozzolanic materials can control the deterioration that might be caused by the alkali-aggregate reactions.
The fine grain size, slow hydration, and, in some cases, particular particle shape add desirable properties to HCC. The heat of hydration is generated more slowly, and, therefore, the temperature of the HCC does not rise so high. One of the most important desirable properties conferred by these materials is the decreased permeability of the resulting HCC. The decreased permeability is not only desirable in and of itself, it also decreases the movement of solutions and diffusion of ions and thus any chemical activity, such as that of alkali reactivity or the activity of invasive deleterious substances. Pozzolanic materials are reported to be effective in increasing the acid resistance of concrete (see Tuthill, 1978a). Natural pozzolans and fly ash should comply with the requirements of ASTM C 618, and GGBFS with ASTM C 989; a separate specification for silica fume is in preparation.
The appearance of a mature concrete in which these materials have been used is generally that of concrete with a low water-cement ratio (see Chapter 9). Pulverized limestone and hydrated lime cannot be positively identified in thin sections of mature HCC. Their presence may be suspected in HCC that appears to have the texture of an HCC with a low water-cement ratio, but they cannot be positively identified. It is impossible to be certain with mature concrete, and without any magnification, if GGBFS or silica fume is present or if the appearances of impermeability are due solely to a low water-cement ratio.
11.2.1 Ground Granulated Blast-Furnace Slag
1. Examine exterior surfaces, broken surfaces, and lapped slices of the HCC. When very young, the pastes containing GGBFS are a characteristic dark bluish green. The chemical reasons for this color in portland blast-furnace clay cements were investigated by Mather (1957), and it was concluded that the development of the color varies from cement to cement and that the coloring agent is in a reduced state, the color being lost on oxidation. Regardless of maturity, if these pastes are kept underwater and not allowed to dry, they will retain this color. As the concrete begins to dry, the surface color fades. If the HCC is sawed or broken while it is only partially dry, it can be seen to be mottled with dark and light places. When it is dry throughout, it will be a uniform light color (see Fig. 11-1). This color is not the same color as ordinary HCC. It is not a gray but rather a very light shade of slightly greenish tan or cream. The mottled appearance of an interior surface of GGBFS-bearing concrete of intermediate age is often of concern to field personnel. It has often been thought that the mixing of the HCC was incomplete. In thin sections examined with the petrographic microscope, the dark-colored areas appear no different from the faded areas and both types of areas seem to have about the same concentration of the GGBFS. If GGBFS was used in the mixture, this mottling is inconsequential and will fade. GGBFS cannot be detected in mature, fully dried concrete with the stereomicroscope.
2. Examine several thin sections of the paste of the specimen with the petrographic microscope. GGBFS can be seen in thin section and is quite distinctive in appearance. It is primarily glass and, therefore, almost completely lacking in birefringence.
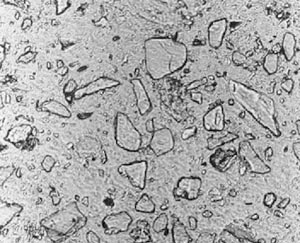
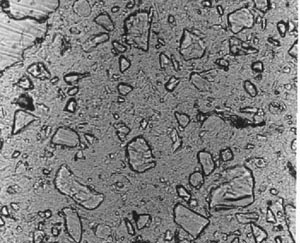
3. Compare the view observed with the views in Figure 11-2. If GGBFS cannot be found by examination of the paste in a thin section, report that it was not detected in the specimen. Figure 11-2 illustrates the appearance of GGBFS at three stages of hydration.
 |
 |
 |
 |
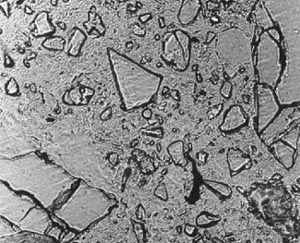 |
Figure 11-2 THIN SECTIONS OF CONCRETE CONTAINING GGBFS. Concentration is 65% of cementitious material. Slag particles are marked with an S. A. At 28 days hydration. Note the angularity of the slag fragments. B. At 56 days hydration. Note the slight rounding of the slag fragments. C. At 6 months hydration. Note the further rounding of the slag fragments.

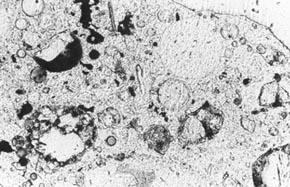
Figure 11-3 ETCHED AREA OF LAPPED SLICE CONTAINING FLY ASH. The partial fly ash particles are indicated by arrows. The particle at the starred arrow is a frothy fly-ash agglomeration. Nearly complete white cenospheres probably filled with froth or a multitude of smaller cenospheres are marked with a W. These whole cenospheres have been almost completely exposed by the etch procedure; some small whole cenospheres may have been lost during the etch procedure. Cupshaped portions of cenospheres are marked with a C.
 |
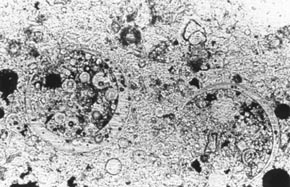 |
Figure 11-4 FLY ASH IN THIN SECTION OF HCC. As seen with petrographic microscope. A. Fly ash particles are indicated by arrows. Note the broken fly ash particle that is composed of dark and light glass. B. Cenospheres of fly ash filled with smaller cenospheres.
1. Examine a lapped slice of the HCC with a stereomicroscope. Fly ash is usually of quite varied particle sizes, and the larger particles can be seen with the stereomicroscope. Most of the individual particles of fly ashes approved for use in HCC are colorless to white. Some of the larger particles are hollow spheres: cenospheres. The walls of the cenospheres are frequently thin enough and the cenospheres large enough to be mistaken for entrained air voids (see Walker, 1983). Sometimes, little dark (black or brown) balls of fly ash (sometimes hollow) can be seen; sometimes, agglomerations of fine fly-ash froth are present (see Fig. 8-6).
2. To detect all the particles of fly ash and distinguish them from air voids, etch the slice or a portion of the slice, rinse, blot, allow to dry, and examine with the stereomicroscope (see 5.2.3). Observe the acid-resistant glass walls of any fly ash particles (see Fig. 11-3). These glass walls are very thin but can be seen with low-angle illumination projecting above the paste that has been etched away. Air voids will not have such projecting walls.
3. Examine thin sections with the petrographic microscope (see Fig. 11-4).
4. If fly ash cannot be detected on an etched lapped slice and cannot be found in the thin section, report that it could not be found in the specimen.
Silica fume cannot be detected by any of the methods commonly in use in the average concrete petrography laboratory. Silica fume has such small particles and is so lacking in birefringence that its presence cannot be detected even in thin section at 400X. Report that the presence of silica fume cannot be detected by the laboratory. (Laboratories that have the capability of examining specimens with a scanning electron microscope can detect and photograph this material.)