U.S. Department of Transportation
Federal Highway Administration
1200 New Jersey Avenue, SE
Washington, DC 20590
202-366-4000
Federal Highway Administration Research and Technology
Coordinating, Developing, and Delivering Highway Transportation Innovations
 |
| This report is an archived publication and may contain dated technical, contact, and link information |
|
Publication Number: FHWA-RD-01-163 Date: March 2002 |
Previous | Table of Contents | Next
Whether a concrete pavement will be affected by MRD is a function of many factors, including the constituent materials used (aggregate, cement, additives, and so on), the location (maritime or inland), the climatic conditions (temperature, moisture), and the presence of external agents (such as roadway deicing chemicals or sulfates in groundwater). In general, the development of MRD can be attributed to either physical or chemical mechanisms, although the two often act together to initiate and propagate distress. The large number of factors and their complex interactions make it difficult in many cases to determine a single "cause" of the observed pavement distress, as more than one mechanism may be at work.
MRD is generally manifest on the pavement surface as fine cracking or as material degradation such as scaling or spalling. The distress may be isolated in the vicinity of joints or cracks or distributed over the pavement surface. Discoloration of the concrete, sometimes referred to as staining, is also a common feature, as is the observation of exudate in cracks. In some instances, concrete expansion may occur, resulting in blowups at joints and displacement of fixed structures. Depending upon the type of distress and the environment to which the pavement is exposed, these distresses may occur as soon as a few years following construction. In other cases, noticeable pavement deterioration will not be observed until much later in the pavement’s service life.
MRD has received greater attention in recent years, in part because of an increased recognition of the importance of material durability in the performance of concrete pavements. Whereas previously it was commonly assumed that sufficient concrete strength ensured durability, it is now recognized that concrete mixtures must be designed for both strength and durability. In addition, it is common today to extend the anticipated concrete pavement design life well beyond the 20 years that was common in the past. This heightens the need for concrete durability to ensure satisfactory pavement performance for the desired 40 to 60 years of service.
Interest in concrete durability has existed almost as long as concrete has been used. Innumerable conferences and thousands of publications have focused on various aspects of concrete durability. Contrary to popular belief, durability is not an intrinsic property of the concrete. One of the most authoritative compilations of this vast amount of information is the Guide to Durable Concrete [American Concrete Institute (ACI) 1992a], which cites over 250 references. In this guide, the "durability of hydraulic-cement concrete is defined as its ability to resist weathering action, chemical attack, abrasion, or any other process of deterioration." In a recent Transportation Research Circular, entitled the Durability of Concrete [Transportation Reseach Board (TRB) 1999], durable concrete is "concrete that in the particular environment of service resists the forces in that environment that tend to cause it to disintegrate without requiring excessive effort for maintenance during its service life." The second definition is more descriptive, as durability is linked not only to the properties of the concrete, but also to the service conditions to which it is subjected during its service life. This concept is captured in the statement that "a given concrete with a given set of properties will endure without noticeable change for centuries or even millennia in one environment and be reduced to fragments in a few years or even a few months in another" (TRB 1999). This linkage between concrete properties and the environment is key to defining what constitutes durability and thus the occurrence of MRD.
In this project, only MRD that commonly affects concrete pavements has been considered. In contrast to the range of deterioration mechanisms considered in the Guide to Durable Concrete (ACI 1992a), some distress mechanisms were not included in this study in recognition that they will only rarely, if ever, affect concrete pavements (acid attack and seawater exposure, for example). Others were not included because the primary cause of deterioration is not directly a result of the interaction between the concrete and environment factors (e.g., moisture, temperature, or external agents). Abrasion is one such distress included in the Guide to Durable Concrete that is not included in this study, since its incidence is primarily linked to the use of tire chains and studded snow tires, which "cause considerable wear to concrete surfaces, even where the concrete is of good quality" (ACI 1992a). Abrasion is thus considered to be primarily related to the loading condition, and is not directly attributable to the interaction between the material and the environment.
As discussed, distresses that are directly related to the interaction between the concrete materials used to construct a pavement and the environment are referred to as MRD. The development of MRD can be attributed to either physical or chemical mechanisms, although the two types of mechanisms often act together to bring about the development of distress. Furthermore, MRD due to multiple causes may develop simultaneously, thereby complicating the determination of the exact cause(s) of material failure.
The following MRD types are considered in this study:
The first three MRD types listed have been categorized as being caused by physical mechanisms and the remaining five by chemical mechanisms. Although this list presents eight distinct MRD types, the complexity of the physical and chemical interactions involved in the development and manifestations of some types of MRD, as well as gaps in the current knowledge base, often prevent absolute identification of the primary mechanism of distress. Also, the fact that more than one distress mechanism may be at work at the same time further complicates the identification process. And finally, it is noted that there are less common occurrences of other types of MRD that are not listed, but may also play a role in concrete pavement deterioration.
Table 1-1 summarizes these distresses, along with general information on their causes, appearance, and prevention; a description of these distresses is provided below. A more detailed description of the distress mechanisms is presented in Appendix A of Volume 1 of the Final Report.
Deterioration Due to Physical Mechanisms
Freeze-Thaw Deterioration of Hardened Cement Paste
Freeze-thaw deterioration of hardened cement paste is caused by the deterioration of saturated cement paste under repeated freeze-thaw cycles. Currently, there is no consensus on the exact mechanisms responsible for internal damage resulting from freeze-thaw action. The most widely accepted theories consider the development of internal tensile stress as a result of either hydraulic pressures, osmotic pressures, or a combination of the two during freezing.
In early efforts to understand freeze-thaw action in concrete, Powers (1945) attributed freeze-thaw damage to excessive hydraulic pressures produced from the expansion of ice. It was proposed that, as ice forms in the pore system, the resulting 9 percent volume expansion causes the surrounding unfrozen water to be expelled from the freezing sites. Depending on the nature of the pore system, excessive internal stresses can develop from hydraulic pressures that are incurred due to resistance to this flow.
More recent theories (Powers 1975) consider osmotic potential to be the primary cause of excess internal stress. As pure water in the larger pores freeze, the liquid remaining in the pores becomes a more concentrated ionic solution. To maintain thermodynamic equilibrium, the less concentrated solution in the surrounding paste is drawn to the freezing sites. If adequate air void space is available, all of the freezable water will eventually diffuse to the freezing sites inside the air voids, reaching a state of equilibrium. If the air void space is inadequate, equilibrium cannot be reached and osmotic pressures sufficient to fracture the paste can result.
Deterioration of the cement paste due to freeze-thaw damage manifests itself in the form of scaling, map cracking, or severe cracking, spalling, and deterioration, commonly initiating at joints and free edges where moisture is more readily available. The addition of an air-entraining agent (an admixture that stabilizes a system of microscopic bubbles in the concrete) is an effective means of preventing this deterioration. This is commonly a top down distress with fractures running nominally parallel with the pavement surface, decreasing in number with depth.
Table 1-1. Summary of key MRDs.
Type of |
Surface Distress Manifestations and Locations |
Causes/ |
Time of |
Prevention or Reduction |
MRD Due to Physical Mechanisms |
||||
Freeze-Thaw Deterioration of Hardened Cement Paste |
Scaling, spalling or map-cracking, generally initiating near joints or cracks; possible internal disruption of concrete matrix. |
Deterioration of saturated cement paste due to repeated freeze-thaw cycles. |
1 to 5 years |
Addition of air-entraining agent to establish protective air-void system. |
Deicer |
Scaling or crazing of the slab surface with possible alteration of the concrete pore system and/or the hydrated cement paste leading to staining at joints/cracks. |
Deicing chemicals can amplify freeze-thaw deterioration and may interact chemically with cement hydration products. |
1 to 5 years |
Provide minimum cement content of 335 kg/m3, limit water–cement ratio to no more than 0.45, and provide a minimum 30-day “drying” period after curing before allowing the use of deicers. |
Freeze-Thaw Deterioration of Aggregate |
Cracking parallel to joints and cracks and later spalling; may be accompanied by surface staining. |
Freezing and thawing of susceptible coarse aggregates results in fracturing and/or excessive dilation of aggregate. |
10 to 15 years |
Use of non-susceptible aggregates or reduction in maximum coarse aggregate size. |
MRD Due to Chemical Mechanisms |
||||
Alkali–Silica |
Map cracking over entire slab area, usually with exudate, and accompanying expansion-related distresses (joint closure, spalling, blowups). |
Reaction between alkalis in the pore solution and reactive silica in aggregate resulting in the formation of an expansive gel and the degradation of the aggregate particle. |
5 to 15 years |
Use of nonsusceptible aggregates, addition of pozzolans to mix, limiting total alkalis in concrete, minimizing exposure to moisture, addition of lithium compounds. |
Alkali–Carbonate Reactivity (ACR) |
Map cracking over entire slab area and accompanying pressure-related distresses (spalling, blowups). |
Expansive reaction between alkalis in pore solution and certain carbonate/dolomitic aggregates which commonly involves dedolomitization and brucite formation. |
5 to 15 years |
Avoid susceptible aggregates, significantly limit total alkalis in concrete, blend susceptible aggregate with quality aggregate or reduce size of reactive aggregate. |
External |
Fine cracking near joints and slab edges or map cracking over entire slab area, ultimately resulting in joint or surface deterioration. |
Expansive formation of ettringite that occurs when external sources of sulfate (e.g., groundwater, deicing chemicals) react with the calcium sulfoaluminates. |
3 to 10 years |
Use w/c below 0.45, minimize tricalcium aluminate content in cement, use blended cements, use pozzolans. |
Internal |
Fine cracking near joints and slab edges or map cracking over entire slab area. |
Formation of ettringite from internal sources of sulfate that results in either expansive disruption in the paste phase or fills available air-voids, reducing freeze-thaw resistance. |
1 to5 years |
Minimize internal sources of slowly soluble sulfates, minimize tricalcium aluminate content in cement, avoid high curing temperatures. |
Corrosion of Embedded Steel |
Spalling, cracking, and deterioration at areas above or surrounding embedded steel with rust present. |
Chloride ions penetrate concrete, resulting in corrosion of embedded steel, which in turn results in expansion. |
3 to 10 years |
Reduce the permeability of the concrete, provide adequate concrete cover, protect steel, or use corrosion inhibitor. |
Deicer Scaling/Deterioration
Deicer scaling/deterioration is typically observed as scaling or crazing of the slab surface due to the repeated application of deicing chemicals. Although the exact causes of deicer scaling are not known, it is commonly believed to be primarily a physical attack. The primary mechanisms considered in the physical deterioration models are High thermal strains produced when a deicer melts ice and/or high osmotic pressures induced when relatively pure surface water attempts to equalize highly concentrated salt solutions present in the concrete (Mindess and Young 1981; Pigeon and Plateau 1995). It has also been speculated that pressure exerted by salt crystallization in voids is a contributing factor(Hansen 1963). Recent studies suggest that chemical alteration of the cement paste may also be occurring, resulting in dissolution of calcium hydroxide, coarsening of the concrete pore system and, potentially, the formation of deleteriously expansive compounds. This occurrence is commonly observed as staining and deterioration in the vicinity of joints.
Deicer scaling/deterioration is more likely to occur in concrete that has been over-vibrated or improperly finished, actions that create a weak layer of paste or mortar either at or just below the surface (Mindess and Young 1981). This weakened layer is more susceptible to hydraulic pressures. Even adequately air-entrained concrete can be susceptible to the development of salt scaling. Recommendations for the prevention of salt scaling include providing a minimum cement content of 335 kg/m3, limiting the water–cement ratio (w/c) to a maximum of 0.45, providing adequate curing, and providing an absolute minimum of 30 days of environmental exposure before allowing the application of deicing chemicals [American Concrete Pavement Association (ACPA) 1992].
Freeze-Thaw Deterioration of Aggregate
Freeze-thaw deterioration of aggregate is a distress associated with the freezing and thawing of susceptible coarse aggregate particles in the concrete. This phenomenon is commonly referred to as D-cracking in pavements [Strategic Highway Research Program (SHRP) 1993], and aggregates are typically identified as being D-cracking susceptible. Such aggregates either fracture and/or dilate as they freeze, resulting in cracking of the surrounding mortar. It has also been hypothesized in some cases that the expulsion of water during freezing contributes to dissolution of soluble paste components, such as calcium hydroxide, in the interfacial zone. Key aggregate properties related to susceptibility are composition, pore structure, sorption, and size (Schwartz 1987). Most susceptible aggregates are of sedimentary origin and are most commonly composed of limestone, dolomite, or chert (Stark 1976).
Freeze-thaw deterioration of aggregate is initially visible as a series of fine cracks generally running parallel to joints, cracks, or free edges in the slab. Deterioration commonly starts near the bottom of the concrete slab where excess moisture accumulates. As the number of freeze-thaw cycles increases, spalling and deterioration of the cracks will occur. A dark staining due to calcium hydroxide or calcium carbonate residue generally precedes and accompanies the cracking, often in an hourglass shape on the pavement surface at affected joints and cracks.
Air entrainment of the cement paste does not prevent the development of D-cracking. The best means of preventing this distress is by prohibiting the use of susceptible aggregate, although reducing the maximum size of the susceptible coarse aggregate has been shown to be effective in reducing freeze-thaw deterioration of aggregate in many instances.
Deterioration Due to Chemical Mechanisms
Alkali–Silica Reactivity (ASR)
Alkali–silica reactivity (ASR) is most commonly associated with undesirable chemical reactions between alkalis in the cement paste (commonly reported as percent Na2O plus 0.658 x percent K2O) and the reactive siliceous components of susceptible aggregates. It is the concentration of the hydroxyl ion in the concrete pore solution that is of interest, which is related to the alkali-content (ACI 1998). The product of the reaction is a gel that significantly expands in the presence of moisture, destroying the integrity of the weakened aggregate particle and the surrounding cement paste. An irregular, map-like cracking ultimately develops, most often over the entire slab area (with cracks generally less than 50 mm deep). ASR can also lead to internal horizontal cracks at greater depths within the slab. Upon continued expansion, joint spalling, blowups, shoving of fixed structures, and other pressure-related distresses in the pavement can occur. A handbook depicting ASR distress in pavements and highway structures is available to aid in its identification (Stark 1991).
The chemical reactions occurring during the development of ASR are very complex, but three basic conditions are needed in order for ASR to occur (Farny and Kosmatka 1997):
The concrete pore solutions are primarily alkali hydroxide solutions of high concentration, which react readily with reactive forms of silica (Stark et al. 1993). As the aggregate reactivity increases, gel reaction products can be formed with lesser concentrations of alkali (Farny and Kosmatka 1997). As the alkalinity (and the pH) of the pore solution increases, the potential for alkali-silica reaction increases as even more stable forms of silica become susceptible to attack. The presence of moisture allows migration of alkali ions to reaction sites and the resulting gel absorbs moisture, leading to expansion (Stark et al. 1993). Relative humidity levels above 80 percent indicate that moisture is sufficiently available for absorption by ASR gel, and recent research has shown that pavement concrete in all climates likely will be sufficiently damp to continuously support expansive ASR (Stark et al. 1993).
Other factors influencing the development of ASR include the total alkali content of the concrete, the presence of external sources of alkalies (e.g., chemical deicers), repeated cycles of wetting and drying, and temperature (Farny and Kosmatka 1997, ACI 1998). A more detailed description of the ASR reaction process is provided by Helmuth (1993).
The gel that is produced through the reaction appears as a glassy-clear or white powdery deposit within reacted aggregate particles, although it is not always visible to the naked eye (Stark et al. 1993). The presence of the reaction product (which is an alkali–calcium–silica–hydrate gel) does not always coincide with distress, and thus gel presence by itself does not necessarily indicate destructive ASR (Farny and Kosmatka 1997).
Common aggregate types containing reactive silica components include opaline or chalcedonic cherts, siliceous limestones, rhyolites and rhyolitic tuffs, dacites and dacite tuffs, andesites and andesite tuffs, and phyllites (Dolar-Mantuani 1982; Neville 1996). An excellent summary of alkali-silica reactive aggregates is presented in the ACI State-of-the-Art Report on Alkali-Aggregate Reactivity (ACI 1998). The rate of the reaction will vary considerably among aggregates, with some undergoing a complete reaction within a matter of weeks and others requiring many years to produce noticeable effects (Helmuth 1993).
A variety of approaches have been tried to prevent or minimize the development of ASR, with mixed success. In new concrete designs, the use of low-alkali portland cement (with an alkali content less than 0.60 percent Na2O equivalent) has been used successfully on slightly to moderately reactive aggregates (Farny and Kosmatka 1997). Some international agencies limit the alkali content of the concrete, accounting for the cement factor and other internal sources of alkalis (ACI 1998). The addition of fly ash has also been shown to control ASR, although this is strongly dependent upon the type of fly ash, its alkali content, chemical composition, and dosage rate (Farny and Kosmatka 1997). Other types of finely divided materials can also be effective including silica fume, ground granulated blast furnace slag, and natural pozzolans (ACI 1998). Finally, the addition of ASR-inhibiting compounds (e.g., lithium hydroxide) has been shown to be effective on highly reactive aggregates (Stark et al. 1993).
For in-service concrete displaying ASR, no definitive method has been identified that is completely successful in stopping the distress mechanisms. The application of silane sealers, methacrylate, and lithium hydroxide have all been tried, with monitoring of the effectiveness of these procedures ongoing (Stark et al. 1993).
Alkali–Carbonate Reactivity (ACR)
Alkali–carbonate reactivity (ACR) is another distress caused by an undesirable chemical reaction between the pore solution and aggregate; in this case, the reaction is between the alkalis in the pore solution and certain dolomitic carbonate aggregates containing a characteristic reactive texture of dolomite rhombs in a clayey fine-grained matrix. Although the mechanisms for ACR are not as well understood as those for ASR, it has been established that dedolomization occurs, that is, the decomposition of dolomite into calcium carbonate and magnesium hydroxide, which is accompanied by expansion. This expansion may be due to a combination of migration of alkali ions and water molecules into the restricted space of the fine-grained matrix surrounding the dolomite crystal, migration of these materials into the crystal, and the growth and arrangement of the dedolomitization products, especially brucite (Farny and Kosmatka 1997, ACI 1998). Other factors influencing the development of ACR include maximum size of the reactive aggregate (rate/degree of expansion decreases with decreasing aggregate size) and pore solution alkalinity (increasing pH levels increase the potential for alkali–carbonate reactions) (Farny and Kosmatka 1997; ACI 1998).
Similar to the expansive pressures that are developed in ASR distress, the expansive pressures developed by ACR also result in map-like cracking on the pavement surface and accompanying expansion-related distresses (spalling, blowups). Avoiding the use of susceptible aggregates is the one sure way of avoiding ACR, but either diluting susceptible aggregates with non-susceptible aggregates or reducing the maximum size of susceptible aggregates can minimize the deleterious effect of the reaction (ACI 1998). Limiting the alkali content in the cement is another method employed to prevent ACR, but the alkali content must be lower than that typically used to prevent ASR (ACI 1998). However, unlike ASR, pozzolans are not effective in controlling alkali–carbonate reaction (Farny and Kosmatka 1997; ACI 1998).
External Sulfate Attack (ESA)
External sulfate attack (ESA) results from chemical reactions that occur when concrete is exposed to penetrating external sulfate ions (present in groundwater, soil, deicing chemicals, etc.). Although the mechanism of sulfate attack is complex, it is primarily thought to be caused by two chemical reactions: 1) the formation of gypsum through the combination of sulfate and calcium ions, and/or 2) the formation of ettringite through the combination of sulfate ions and hydrated calcium aluminate (ACI 1992a, DePuy 1994). In either case, the formation of the reaction product leads to an increase in solid volume. In the former case, expansion due to gypsum formation may not be destructive, but gypsum has little cementing properties and thus the concrete loses integrity (DePuy 1994). On the other hand, the expansive pressures exerted by ettringite formation can be very destructive.
In concrete pavements, deterioration due to external sulfate attack typically first appears as cracking near joints and slab edges, generally within a few years of construction. Fine longitudinal cracking may also occur parallel to longitudinal joints. Steps taken to prevent the development of distress due to external sulfate attack include minimizing the tricalcium aluminate content in the cement or reducing the quantity of calcium hydroxide in the hydrated cement paste through the use of pozzolanic materials. It is also recommended that a w/c ratio less than 0.45 will help mitigate external sulfate attack (ACI 1992a).
Internal Sulfate Attack (ISA)
Internal sulfate attack (ISA) is a potential pavement distress similar in many ways to external sulfate attack, except that the source of the sulfate ions is internal. Internal sources of sulfate include slowly soluble sulfate contained in clinker, aggregate, and admixtures (such as fly ash) or as a result of decomposition of primary ettringite during early hydration, primarily associated with high curing temperatures. This particular distress has elicited considerable debate among concrete material experts regarding the specific mechanisms of distress and the precise role of ettringite in its development. Sometimes called delayed ettringite formation (DEF) distress or secondary ettringite formation (SEF) distress, it is referred to here as internal sulfate attack to distinguish the source of sulfate ions. For consistency and clarification, the following definitions are offered regarding the various forms of ettringite (based on Erlin 1996a):
Ettringite—a high-sulfate calcium sulfoaluminate mineral (3CaO • Al2O3 • 3CaSO4 • 32H2O).
Primary Ettringite—ettringite formed by reaction of sulfate and aluminate ions during early hydration of hydraulic cement either as a normal process for portland cement or as the expansive process for expansive cement.
Secondary Ettringite—ettringite commonly formed in available void space by precipitation from solution of either primary or delayed ettringite.
Delayed Ettringite—ettringite formed by reaction of sulfate and aluminum ions in concrete, mortar, or grout that has hardened and developed its intended strength; the source of the sulfate ions is from within the concrete.
It is believed that both SEF and DEF are forms of internal sulfate attack, but result for different reasons. SEF is commonly a product of concrete degradation, characterized by the dissolution and subsequent precipitation of ettringite into available void space and in pre-existing microcracks. SEF can occur in concrete that is sufficiently permeable and saturated, allowing the dissolution and precipitation process to occur. Although most experts agree that secondary ettringite formation will not generate sufficient expansive pressures to fracture healthy cement paste or mortar, its presence in the air void structure may limit the ability of the paste to resist freeze-thaw deterioration (Ouyang and Lane 1999). Thus, concrete that appears to be suffering paste freeze-thaw deterioration may have originally had an adequate air void system that has been compromised by SEF. Yet there remains considerable debate as to whether the SEF is the cause of distress or only present as a result of another deterioration mechanism that disrupted the paste sufficiently to encourage dissolution and precipitation of the ettringite (Detwiler and Powers-Couche 1999).
DEF, on the other hand, can lead to destructive expansion within the paste, resulting in microcracking and separation of the paste from aggregate particles. DEF is most often associated with steam curing. At elevated temperatures (current research suggests a minimum temperature of 65 °C to 80 °C, with many citing 70 °C [Scrivener 1996; Thaulow et al. 1996a; Klemm and Miller 1999]), primary ettringite will not form properly. After the concrete has cured and temperatures are reduced to ambient conditions, sulfates and aluminate phases in the paste may then react to form expansive ettringite, disrupting the concrete matrix. Because this phenomenon is most closely associated with steam curing, it is still speculative whether cast-in-place pavements can experience the temperatures necessary to produce DEF. It has also been suggested that internal sulfate attack might occur due to internal sources of sulfate that become available after the paste has hardened. Possible sources include either slowly soluble sulfates or sulfur compounds in the clinker or fly ash that only become available during continued long-term hydration. Another internal source of sulfates might be from aggregates (Johansen and Thaulow 1999). In these cases, the internal sulfate attack is not from DEF, but instead from excess sulfates in the concrete mixture, which result in paste expansion along similar lines as external sulfate attack.
The manifestation of internal sulfate attack in many concrete structures is characterized by a series of closely spaced, tight map cracks with wide cracks appearing at regular intervals. Microscopically, paste expansion due to DEF can be identified by gaps in the paste around aggregates with widths proportional to the particle size (Johansen and Thaulow 1999).
Only recently have researchers started investigating internal sulfate attack as a potential pavement distress mechanism, and possible means of prevention are still being explored. Recommendations from a recently completed study indicate that limiting the sulfate content of cement and fly ash may assist in preventing ISA, but that it is impossible to recommend a specification at this time (Gress 1997). Other studies suggest that limiting sulfate content is only an issue if high curing temperatures are to be expected (Scrivener 1999). The extent of the problem in pavements appears to be limited at this point, but additional research is necessary to determine both the extent of internal sulfate attack and feasible strategies to prevent its occurrence in new pavements.
Corrosion of Embedded Steel
Corrosion of embedded steel appears as rust colored staining, spalling, cracking, and associated deterioration of the concrete above or surrounding the steel affected by active corrosion. Steel corrosion is accelerated in the presence of chloride ions (which can come from a chloride-based accelerator added to the mix, deicing salts, or seawater) that break down the passivity film that protects embedded steel from corrosion.
Corrosion of embedded steel is controlled by prohibiting chloride additions to fresh concrete, decreasing permeability of the concrete, providing adequate concrete cover over the steel, coating steel with a protective layer, application of a protective coating on the concrete surface to prevent penetration of chlorides and moisture, and suppression of the electrochemical process at the steel surface.
Due to the complexity of the distress mechanisms at work, the specialized laboratory methods employed, and the lack of standardization in the evaluation process, it became apparent that guidance was needed to assist SHAs in the evaluation of MRD affected concrete pavements. As part of this project, the following three guidelines were developed to provide pavement engineers and field and laboratory personnel with a systematic procedure for the identification, evaluation, treatment, and prevention of MRD in PCC pavements:
These guidelines have been prepared with the practitioner in mind, and it is expected that the application of a standardized, systematic approach to the overall data collection, analysis, and interpretation process will provide an accurate assessment of the type of distress(es) present and their cause(s). Furthermore, information collected in accordance with the guidelines will prove useful as additional research and investigations are conducted on MRD affected pavements.
Need for Advisory Guidelines
The guidelines have been prepared to assist pavement engineers and field and laboratory personnel in the identification, evaluation, treatment, and prevention of MRD in concrete pavements. Investigations of concrete pavement deterioration have been conducted for years by numerous agencies, laboratories, and institutions, each with their own set of guidelines and testing procedures. Some SHAs have studied MRD in great detail, establishing unique testing protocols and material specifications. Other agencies, however, are not familiar with these procedures. The American Society for Testing and Materials Standard Practice for Petrographic Examinations of Hardened Concrete (ASTM C 856) provides an excellent outline of procedures for petrographic evaluation of concrete, but is focused strictly on petrographic means and is not specifically oriented toward the evaluation of MRD affected concrete pavements. In summary, there is a lack of information on the subject, and where information exists, there is little standardization regarding nomenclature, test methods, and identification procedures. The lack of standardization in test methods and data collection procedures makes it difficult to reach consensus on diagnoses and to analyze the performance of concrete pavements on a national basis.
Purpose and Objectives of Guidelines
The overall purpose of the guidelines is to provide a standardized approach for field evaluation, sampling and handling, and laboratory procedures for use in identifying MRD in pavements. Furthermore, these guidelines will provide useful information on effective measures for the economical preservation of PCC pavements identified as suffering MRD and guidance for the construction of long-lasting PCC pavements. It is expected that these guidelines will assist in the free flow of information between various agencies, laboratories, and institutions so that all benefit from the findings of each. Figure 1-1 illustrates the flow of the three guidelines involved in the identification, treatment, and prevention of MRD.
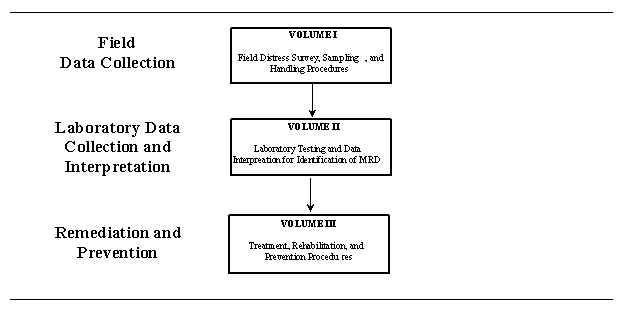
Figure 1-1. Flow of guidelines for identification, treatment, and prevention
of MRD.
Previous | Table of Contents | Next