U.S. Department of Transportation
Federal Highway Administration
1200 New Jersey Avenue, SE
Washington, DC 20590
202-366-4000
Federal Highway Administration Research and Technology
Coordinating, Developing, and Delivering Highway Transportation Innovations
| REPORT |
| This report is an archived publication and may contain dated technical, contact, and link information |
|
| Publication Number: FHWA-HRT-11-045 Date: November 2012 |
Publication Number: FHWA-HRT-11-045 Date: November 2012 |
The United States produces hundreds of millions of tons of hot mix asphalt (HMA) each year for pavement construction and maintenance. Although the asphalt weighs less and represents a smaller proportion of the HMA mixture, the liquid asphalt binder component is more costly than the stone aggregate component, translating to billions of dollars spent annually. Asphalt binders for HMA are purchased, graded, and verified using the SUperior PERforming Asphalt PAVEment (Superpave®) performance grade (PG) system developed by the Strategic Highway Research Program (SHRP).
The aim of the Superpave® PG system and asphalt binder specifications is to ensure acceptable performance of flexible asphalt pavements in three distinct temperature or seasonal regimes, each associated with a different distress. The assurance of acceptable performance comes with the following requirements:
State transportation agencies specify PG binder using specifications adopted by the American Association of State Highway and Transportation Officials (AASHTO). AASHTO M 320, Standard Specification for Performance-Graded Asphalt Binder, assigns three temperature grades to a particular asphalt binder using the following three tests:(2)
Both AASHTO T 313 and AASHTO T 314 measure material properties intended to control low-temperature thermal cracking performance.(3,4) This is not within the scope of this research. AASHTO T 315 measures material properties intended to control both high-temperature rutting and intermediate-temperature fatigue cracking distresses. The rheological properties of asphalt binders characterized using a dynamic shear rheometer (DSR) are the viscoelastic (complex) shear modulus, |G*|, and viscoelastic phase angle,δ. Temperature and rate of loading affect these rheological properties, which is why they are considered viscoelastic in nature. Increasing temperature decreases asphalt binder stiffness while increasing the viscoelastic phase angle and vice versa. Decreasing the rate of loading has the same effect as increasing temperature. SHRP’s Asphalt Research Program recommended combinations of the shear modulus and phase angle as specification criteria for rutting and fatigue cracking.(6)
SHRP was initiated to increase the life of pavements and decrease life-cycle costs and maintenance requirements. Asphalt research focused on delivering two products: a performance-based binder specification and an asphalt aggregate mixture design and analysis system. The research was broken into the following contracts:(7)
To achieve the desired products, the research was broken into four distinct phases. The first phase was conceptualization, identifying candidate physiochemical phenomena in binders and mechanical properties of mixtures that govern asphalt pavement performance. The second phase was definition, defining the asphalt binder properties that would be validated against laboratory accelerated mixture performance tests and, to a lesser degree, with full-scale accelerated pavement testing (APT). These activities were considered a first-stage validation. At the same time, tests suitable for specifications were developed. This phase was followed by the validation phase, during which field performance data were used to complete the first-stage validation of binder and mixture properties that were judged to have a strong effect on pavement performance in the definition phase. This was considered the second-stage validation. The last stage was adoption, where the implementation of binder and mixture specifications begins. Ultimately, the third-stage validation would come from Long-Term Pavement Performance (LTPP) Specific Pavement Study (SPS)-9 test sections.
SHRP contracts A-002A, A-003A, and A-005 had the greatest influence on research and recommendations leading to the current asphalt binder PG specifications. Contract A-002A was tasked with identifying chemical and physical properties of asphalt binders that were associated with performance and developing specification tests for these properties. Contracts A-003A and A-005 supported A-002A to provide validation. Contract A-003A developed standard laboratory asphalt-aggregate mixture tests based on properties identified in A-002A. Contract A-005 provided the basis for criteria and limits to refine asphalt binder and mixture specification tests from field performance. The interaction among contracts is shown graphically in figure 1, which is reproduced from the SHRP Asphalt Research Program strategic plan.(7)
Figure 1. Flowchart. SHRP asphalt strategy.(7)
SHRP contract A-002A was comprehensive and focused on a molecular microstructural chemical model for the asphalt binder, aging and oxidative mechanisms, and physical rheological properties that are the subject of this research. SHRP A-367 describes physical rheological properties for specification tests and presents why various empirical techniques are inferior to fundamental viscoelastic properties, which are the basis for the current Superpave® PG specifications.(6) The primary advantage of fundamental viscoelastic rheological properties of asphalt binder is the ability to account for temperature effects, aging effects, shear rate, and viscosity effects.
SHRP A-369 explains why and how the fundamental viscoelastic rheological properties were developed and chosen and, importantly, describes limits and criteria for those properties in the current practice found in AASHTO T 315.(8,5)
With respect to fatigue cracking, the SHRP researchers responsible for developing specification tests were aware of the complicated fundamental fatigue and fracture phenomena associated with asphalt cracking. These include stress concentrations found at the leading edge of crack tips and mathematics regarding the propagation of those cracks as well as the inelastic conditions that influence fracture mechanics theories. The researchers identified the presence of plasticity and viscoelasticity and the energy dissipation at intermediate temperatures where fatigue cracking is assumed the dominant distress. Relationships between fatigue and fracture did not exist because the profession lacked a convenient test for asphalt binder fatigue. Fracture was explicitly explored with direct tension (DT) fracture tests for low-temperature cracking. Master curves of failure stress and failure strain over wide ranges of temperature and strain rate were developed as a possible means to extend DT fracture testing from low temperatures (thermal transverse cracking) to intermediate temperatures (fatigue, alligator cracking, etc.). However, doing so would require a transition from the brittle state at low temperatures to a combined ductile-brittle state at warmer temperatures.
Explicit justification for a practical surrogate for advanced fundamental tests was that fatigue tests are too lengthy for practical specifications and fatigue phenomena in the field occur over a broad range of temperatures and stress levels depending on traffic and location within the pavement structure. Fundamental fatigue tests had to be conducted to support the choice of a surrogate test. SHRP researchers utilized reduced-scale three-point bending beam geometries of composite aluminum metal and asphalt binder. Strain magnitudes of the beam undergoing fatigue were between 5,200 and 1,400 microstrain (0.52 and 0.14 percent), and the temperature range was -22 to 32 °F (-30 to 0 °C). It is interesting to note that such classical cyclic fatigue characterization tests are contemporarily conducted in parallel plate geometry using DSR at more intermediate temperatures, as discussed in later sections of this report. Failure was defined as the appearance of a crack in the asphalt beam.
A number of phenomena were confirmed. First, the larger the induced repeated strain in the fatigue test, the fewer number of cycles to achieve failure. Second, the ratio of the energy lost due to viscoelasticity and fatigue damage in a given cycle compared to the total energy input for a given cycle remains constant as cycles increase during the fatigue test but increase dramatically at the end when damage dominates. An example of this from SHRP research is shown in figure 2. The second observation was that energy dissipated is a strong indicator of fatigue damage. Analytical closed-form solutions based on dissipated energy were derived to equate the number of cycles to the strain level and total dissipated energy for an entire fatigue test. Experiment and theory matched reasonably well, as shown in figure 3. DSR instruments were introduced in the SHRP project, and dissipated energy was related to the parameter |G*|× sinδ. This parameter was ultimately selected as the specification controlling fatigue cracking. Justification for the parameter criteria initially set at 435 psi (3 MPa) was made utilizing data from the Zaca-Wigmore road test built in the mid to late 1950s to assist the California Department of Highways in evaluating penetration binder specifications and durability.(9,10) Figure 4 shows the data where sections with larger |G*| × sinδ values exhibited greater amounts of cracking. SHRP researchers utilized this data to justify the limit of 435 psi (3 MPa) for |G*| × sinδ. However, the Federal Highway Administration (FHWA) Asphalt Expert Task Group (ETG) suggested increasing the limit to 725 psi (5 MPa), which can be found in the current practice specification of AASHTO M 320.(2)
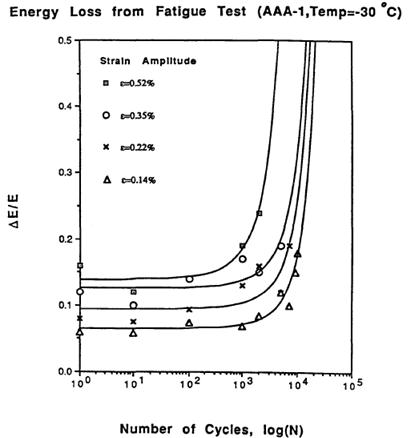
Figure 2. Graph. Dissipated energy during asphalt binder bending beam fatigue.(8)
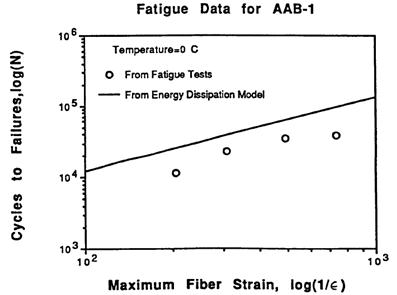
Figure 3. Graph. Relationship between strain level and fatigue life in asphalt binder.(8)
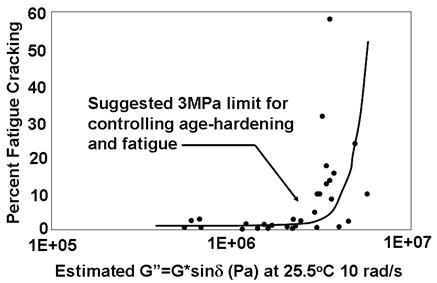
Figure 4. Graph. Zaca-Wigmore test road cracking performance and estimated binder properties.(6)
With respect to rutting specifications and criteria, SHRP A-369 proposed that asphalt binders exhibit both recoverable and irrecoverable components of viscoelastic deformations and that it is the irrecoverable component that contributes to the permanent deformation and rutting observed in full-scale pavements.(8) To explore these phenomena, a unique apparatus and asphalt binder sample test configuration was developed. An actuator provided repeated loading with rest periods to a piston that penetrated a sample of asphalt binder controlled at a fixed temperature. The apparatus measured the recoverable and irrecoverable deformations. The rest period allowed the recoverable deformations to attenuate from the total deformations, leaving behind a permanent or plastic deformation before the next load cycle. The experimental program measured these plastic and permanent deformations on a series of asphalt binders, some unmodified and some polymer modified. The SHRP research found that large permanent deformations grew in a nonlinear fashion. More practical indentation tests were explored as an alternative; however, the material response of interest was not necessarily the plastic deformations but the manner in which stresses relax in asphalt binder from a monotonic indentation. This characterization was done at cooler-than-ideal high-temperature rutting conditions. In other words, the researchers were looking to measure fundamental linear viscoelastic relaxation modulus. All previous mechanical characterization instruments were abandoned as SHRP research adopted DSRs, which generate dynamic data that can be converted to time-domain data and provide the relaxation of interest.
SHRP A-367 indicates that originally, the "viscous component of stiffness" was found not to correlate with mixture data.(6) The type of mixture data and quality of the correlation were not provided in the final reports. Ultimately, the loss compliance was selected as the ideal specification parameter, which could be calculated from the DSR instrument as the shear modulus divided by the sine of the phase angle, |G*|/sinδ. Justification for this parameter shown in figure 5 came from personal communication that correlated non-descript wheel tracking tests of six unknown mixture data points with loss compliance (|G*|/sinδ).
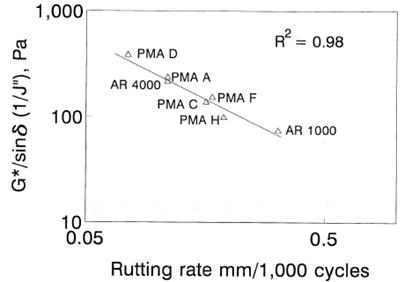
1 Pa = 0.000145 psi
1 mm = 0.039 inches
Figure 5. Graph. SHRP justification for the selection of high-temperature rutting criteria.(7)
Asphalt binder parameters proposed by the SHRP A-002A contract were tested and validated against the extensive and comprehensive laboratory mixture tests conducted by the SHRP A-003A contract. SHRP A-404 and SHRP A-515 provide an exhaustive description of the various tests explored for fatigue characterization and rutting/permanent deformation.(11,12) Ultimately, flexural beam fatigue tests and simple shear testing were selected as the ideal laboratory characterization tests. A summary of the comparison between binder properties and mixture properties is given in SHRP A‑398 and also in a more concise summary by Leahy et al.(13,14)
To evaluate the fatigue binder specification, mixtures from eight SHRP core asphalt binders (unmodified) and two SHRP core aggregates were tested at two air void levels and repeated strain levels. A total of 128 specimens were tested. The experimental design examined the effects of binder type, aggregate type, density, and binder-aggregate and binder-density interactions. Binder type was found to have the strongest influence on fatigue life, as defined by 50 percent reduction in modulus. However, researchers found that other factors were also significant contributors and could not be considered negligible. Comparisons were made between the fatigue life of the mixtures and binders. To eliminate interaction effects from the other variables, four scenarios (the combinations of the two aggregates and two density levels) were analyzed. Cyclic strain level was averaged. An example of one of the SHRP scatter plots for fatigue life versus binder property is shown in figure 6, but figures for dissipated energy and other properties were also provided. Each comparison has eight data points, and Pearson’s correlation coefficient, R, was calculated and is given in table 1. (Note that this is not the coefficient of determination, R2.) The fatigue life, in general, had a good relationship with the logarithmic value of the specification |G*| × sinδ equal to the loss modulus G'', the logarithmic value of the complex stiffness |G*|, and the logarithmic value of the storage modulus G' equal to |G*| × cosδ. For binders with the same specification parameter |G*| × sinδ, stiffness could have as much as twice the fatigue life depending on the aggregate and density. The SHRP researchers noted that the phase angle had little effect and that any of the stiffness relationships could be used. However, it was recognized that modified asphalt could benefit from the sinδ term.
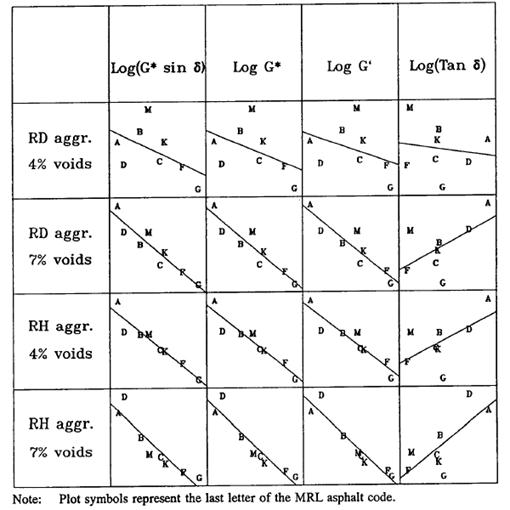
RD = Quarried,100 percent crushed aggregate.
RH = Partially crushed river gravel.
Figure 6. Chart. Comparison between SHRP mixture flexural beam fatigue and asphalt binder rheology.(13)
|
Mix Property |
Flexural Stiffness |
Fatigue Life |
Dissipated Energy |
|---|---|---|---|
|
Aggregate RD, 4 percent air voids |
|||
|
Log (|G*|•sinδ ) |
0.906 |
-0.535 |
-0.32 |
|
Log |G*| |
0.904 |
-0.474 |
-0.241 |
|
Log G' |
0.888 |
-0.401 |
-0.149 |
|
Log(tanδ) |
-0.564 |
-0.156 |
-0.456 |
|
Aggregate RD, 7 percent air voids |
|||
|
Log (|G*|•sinδ ) |
0.905 |
-0.935 |
-0.672 |
|
Log |G*| |
0.909 |
-0.927 |
-0.622 |
|
Log G' |
0.897 |
-0.915 |
-0.568 |
|
Log(tanδ) |
-0.606 |
0.578 |
0.062 |
|
Aggregate RH, 4 percent air voids |
|||
|
Log (|G*| × sinδ ) |
0.951 |
-0.951 |
-0.806 |
|
Log |G*| |
0.946 |
-0.945 |
-0.76 |
|
Log G' |
0.926 |
-0.933 |
-0.707 |
|
Log(tanδ) |
-0.571 |
0.6 |
0.175 |
|
Aggregate RH, 7 percent air voids |
|||
|
Log (|G*| × sinδ ) |
0.952 |
-0.927 |
-0.925 |
|
Log |G*| |
0.935 |
-0.944 |
-0.935 |
|
Log G' |
0.902 |
-0.952 |
-0.935 |
|
Log(tanδ) |
-0.473 |
0.753 |
0.692 |
RD = Quarried 100 percent crushed aggregate.
RH = Partially crushed river gravel.
SHRP conducted an analysis that expanded upon the mixture effects (i.e., air voids and density). The impact of pavement structural configuration (i.e., stiff versus soft or thick versus thin layers) was explored, as well. The results of the beam fatigue tests were used to calibrate the generic model for fatigue life as a function of the inverse of the tensile strain magnitude. Two scenarios were considered: (1) an asphalt concrete (AC) layer on aggregate base over a subgrade and (2) a thicker AC layer on a softer subgrade. The fatigue lives of the fictitious pavements predicted from the equation were compared against the binder properties. The relationships between binder and pavement performance were weaker than the relationships between binder and laboratory test fatigue life shown in table 1 and figure 6. In addition, the trends with lab-mix specimens were opposite the trends with binder. Notably, the generic model for pavement structure fatigue life did not include stiffness, as does the current empirical equation in the National Cooperative Highway Research Program (NCHRP) 1-37A, Mechanistic-Empirical Pavement Design Guide (MEPDG) methodology.(1) The SHRP analysis shows that structural configuration of pavement has a significant contribution to overall performance and, depending on the situation, may override the choice of asphalt binder.
The approach used with respect to rutting was similar to that used for fatigue characterization. Wheel tracking tests and laboratory shear tests were conducted. Sixteen asphalt binders and two aggregates were used to make samples at two air void levels for wheel tracking tests. Nine asphalt binders and two aggregates were used to make samples at two air void contents for the laboratory shear tests. Statistical analysis of variance (ANOVA) was conducted to assess the proportional effect of binder type, aggregate type, density, and the interaction between binder and aggregate. In contrast to the fatigue analysis, there were almost equal contributions from aggregate, binder, and binder-aggregate interaction for the wheel tracking tests but less so for density. Statistical findings were similar for the laboratory repeated shear testing, except with a bit larger error than wheel tracking. As with the fatigue analysis, the laboratory wheel tracking and shear tests were compared to the characteristics of the binder only by separating the data into the four combinations of the two aggregates and two density levels. Comparative plots and correlation calculations of the binder properties against wheel tracking and laboratory repeated shear were conducted similar to the fatigue characterization. Typical results from SHRP are shown in figure 7. Significant scatter was observed, with inconsistent trends at times being positive or negative for a particular scenario of aggregate type, density, and laboratory characteristics. The SHRP researchers concluded that binder properties can be overridden by aggregate properties. One comment from the SHRP researchers was that the tests were conducted at the relatively low temperature of 104 °F (40 °C), and higher temperature tests could emphasize the effect of binder, as in this Transportation Pooled Fund (TPF) research.
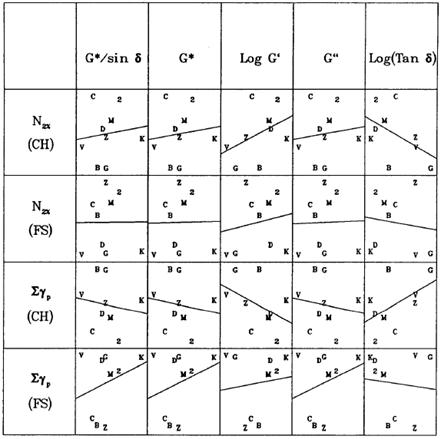
Figure 7. Chart. Example of SHRP repeated shear and shear stiffness of mixtures compared against asphalt binder rheology.(13)
SHRP contract A-005 provided more near-term validation of the selected binder specifications until the results from the LTPP program could yield sufficient data. The research was intended to provide documented field performance data to help set criteria to the parameters determined in SHPR contract A-003. SHRP A-357 provides empirical validation of the binder specifications for load-related distresses fatigue cracking and rutting.(15)
SHRP General Pavement Study (GPS) test sites were used by the researchers. Asphalt binder from 29 sections was extracted and characterized for comparison. The 29 sections came from Alabama, Alaska, Arizona, Arkansas, California, Colorado, Florida, Idaho, Illinois, Kentucky, Maine, Michigan, Mississippi, Nevada, New Jersey, New Mexico, Oklahoma, Tennessee, Utah, and Quebec. Two types of pavement performance prediction software, FLEXPASS and Texas Flexible Pavement System, were used to calculate predicted rutting and cracking distresses based on material properties measured on the mixtures in the laboratory and back-calculated properties from nondestructive techniques. The ratio of measured to predicted distresses was calculated. Ratios larger than 1.0 were categorized as high, and ratios smaller than 1.0 were categorized as low.
The logic for this analysis was to eliminate contributions from supporting layers and emphasize contributions from the AC layer. Binder |G*| was plotted against binder phase angle, the two components of the rutting and fatigue cracking specification parameters. Each point was identified with its corresponding high or low ratio of predicted versus measured distress. The graph for fatigue cracking in the GPS test sections shows no clear trends, with the high and low points interspersed among each other (see figure 8). The quality of the scatter plot is similar for rutting. The conclusion drawn by SHRP A-005 researchers was that no binder specification by itself can explain field performance.
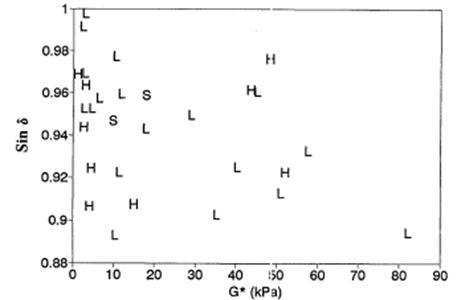
1 kPa = 0.145 psi
Figure 8. Graph. G* versus sinδ binder test values for high and low rates of fatigue cracking.(13)
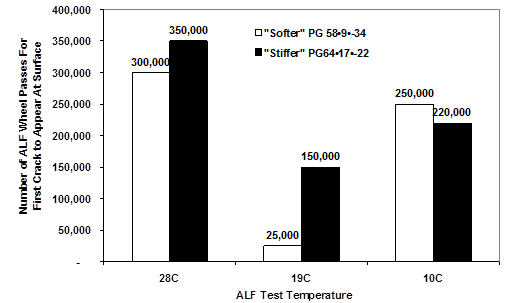
FHWA provided APT to validate the newly developed SHRP binder specifications |G*| × sinδ and |G*|/sinδ.(16–18) Two unmodified asphalts with high, intermediate, and low PG temperature grades of 58, 9, and -34 and 64, 17, and -22 were used in a single mix design with 4- and 8-inch (100- and 200-mm)-thick asphalt layers, respectively. Test pavements were trafficked by an accelerated load facility (ALF) at 50, 66, and 82 °F (10, 19, and 28 °C).
The fatigue experiment was designed to evaluate whether stiffer binders defined by |G*| × sinδ and, thus, stiffer mixtures, performed better in thicker asphalt layers while softer, more compliant binders defined by lower |G*| × sinδ and, thus, softer mixtures, provided better fatigue cracking resistance in thinner asphalt pavements. The experiment also tested whether there was a pessimum temperature for fatigue cracking. In other words, there should be less fatigue cracking at temperate cooler and warmer temperatures than at some critical intermediate temperature.
These fatigue phenomena are partially confirmed in figure 9 and figure 10, where the middle test temperature of 66 °F (19 °C) required fewer passes to produce cracks than did 50 or 82 °F (10 or 28 °C). However, binder |G*| × sinδ only partly explained the performance in the thinner pavement, where strain control phenomena were believed to dominate. This is not a departure from classical pavement engineering. At the intermediate 66 °F (19 °C) temperature for both 4- and 8-inch (100- and 200-mm)-thick pavements, the binder with the stiffer |G*|•sinδ performed better, contrary to the intent of the specification. The researchers suggest that the loss compliance used for high-temperature binder specifications, |G*|/sinδ, is associated with stress control fatigue phenomena and |G*| × sinδ is associated with strain control fatigue phenomena and that stress control |G*|/sinδ should be used to grade fatigue performance for thicker pavements.
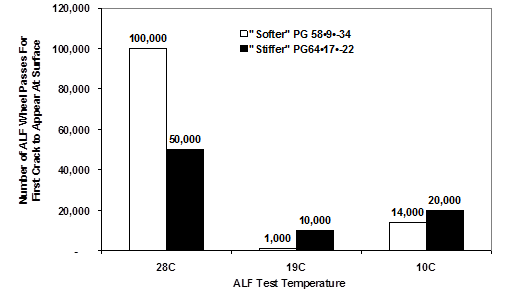
°F = 1.8(°C) + 32
Figure 9. Graph. FHWA APT validation of SHRP binder fatigue specification for 4-inch (100-mm) HMA test sections.(16)
°F = 1.8(°C) + 32
Figure 10. Graph. FHWA APT validation of SHRP binder fatigue specification for 8-inch (200-mm) HMA test sections.(16)
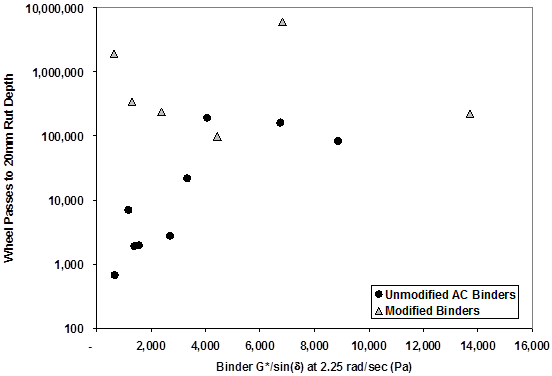
Rutting was evaluated at multiple temperatures between 115 and 169 °F (46 and 76 °C) on five asphalt binders. Three binders were unmodified: AC-5, AC-10, and AC-20, with high-temperature grades of PG58, PG64, and PG70, respectively. Two binders, Novophalt and Styrelf, were modified asphalts with high-temperature grades of PG76 and PG82. The number of wheel passes to achieve 0.8‑inch (20-mm) rut depth was used to assess the validity of the parameter |G*|/sinδ. However, the parameter at a slower frequency of 2.25 radians/s in the DSR was chosen instead of the standard 10 radians/s because of the slow speed of the FHWA ALF.
As shown in table 2, binders were characterized at three temperatures, and test lanes were loaded at those same temperatures, depending on the stiffness of the binders. For the polymer modified asphalts, the rutting was quite low, and thus, power law models were fit to the measured rutting versus wheel pass data to extrapolate to the 0.8-inch (20-mm) rut depth criteria. A very broad range in wheel passes was found and is presented in figure 11 using a semilogarithmic scale.
|
Binder |
Wheel Passes to 20-mm Rut Depth |
Temperature (°C) |
|G*|/sinδ at 2.25 radians/s |
|---|---|---|---|
|
AC-5 (PG58) |
192,000 |
46 |
4,061 |
|
1,950 |
52 |
1,557 |
|
|
670 |
58 |
664 |
|
|
AC-10 (PG64) |
83,000 |
46 |
8,865 |
|
21,720 |
52 |
3,329 |
|
|
1,900 |
58 |
1,384 |
|
|
AC-20 (PG70) |
161,400 |
52 |
6,744 |
|
2,740 |
58 |
2,702 |
|
|
7,000 |
64 |
1,175 |
|
|
Novophalt (PG76) |
6,000,000 |
58 |
6,826 |
|
340,000 |
70 |
1,304 |
|
|
1,900,000 |
76 |
642 |
|
|
Styrelf (PG82) |
220,000 |
58 |
13,710 |
|
98,300 |
70 |
4,435 |
|
|
236,000 |
76 |
2,381 |
1 mm = 0.039 inches
°F = 1.8(°C) + 32
1 Pa = 0.000145 psi
1 Pa = 0.000145 psi
1 mm = 0.039 inches
Figure 11. Graph. Post-SHRP rutting binder validation conducted by FHWA.(18)
The association between the binder parameter and full-scale rutting was fairly weak. However, when considering only the unmodified binders, the trends were correct and reasonable. Additional analyses were conducted by fixing the wheel passes and evaluating the association with rut depth at a fixed number of wheel passes. Aging of the binders was considered because the pavements were tested at different times. Extracted binders were obtained and compared after the ALF tests. The alternative analyses did not change any of the conclusions regarding the poor association between rutting and the binder parameter in modified asphalts.
LTPP SPS-9 experiments were developed to provide validation of the SHRP asphalt PG binder specification and Superpave® volumetric mix design. Three subsections were built in each test section: one for the particular agency’s binder and mix design specification, one for the PG binder specification and Superpave® volumetric mix design, and one for the Superpave® volumetric mix design with a PG binder intentionally chosen incorrectly to achieve failure sooner and capture binder effects. In 2001, an analysis of the SPS-9 test sections was conducted that recognized several caveats.(19) Limitations to the analysis were as follows:
Twenty-six test sections were built between 1992 and 1998, and the data were analyzed in 2001. Low-, medium-, and high-severity cracks were combined, and the standard definition for fatigue cracking of interconnected cracks within the wheel path was followed. Comparisons among the agency design, SHRP PG Superpave®, and SHRP alternate PG sections used the SHRP extent-of-distress bins of normal, moderate, and severe distress categorization.
In total, 78 percent of the 26 sections had no cracking, and 80 percent exhibited nominal rutting. Direct comparison of the extent of distress between sections showed that the agency selection specification had less rutting but more cracking than the SHRP PG and Superpave® sections. Statistical F-tests and t-tests were conducted to evaluate the distresses between the SHRP PG Superpave® sections and the SHRP alternate PG sections to determine whether or not there were any statistical differences in performance when the incorrect PG binder was used. At a 95 percent confidence level, no differences were found between the SHRP PG sections and the alternate PG sections in rutting, wheel path fatigue cracking, and non-wheel path longitudinal cracking. When the material properties data were investigated, a reported 18 of the 26 alternate sections did not correctly choose the "incorrect" binder specification to produce greater rutting and cracking.
NCHRP 9-10, which was launched in 1996, was a landmark study in response to the growing evidence and concerns that SHRP PG binder could not adequately explain performance for modified asphalts.(20) Classes of asphalt binder modifiers were identified in practice and research. These classes were fillers, extenders, polymer elastomers, polymer plastomers, crumb rubber, oxidants, hydrocarbons, process-based, fibers, and antioxidants. Detailed surveys were conducted and summarized the extent to which modifiers were used by highway agencies and the distresses that were targeted. Contractors and suppliers provided experience in terms of the process of modification of simple and complex binders. Complexities arose from issues such as size and content of particles (i.e., crumb rubber), long-term stability, strain sensitivity or nonlinearity, and cyclic or creep sensitivity.
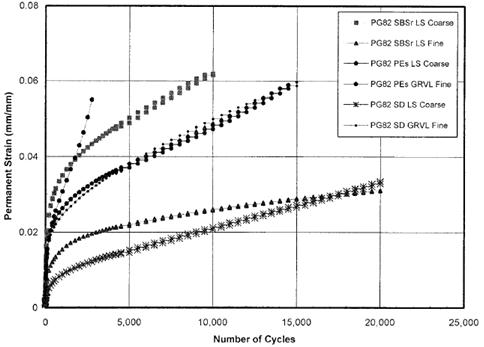
Laboratory tests on asphalt mixtures were conducted in the NCHRP 9-10 research. Repeated shear at constant height (RSCH) tests were conducted on identical mix designs with different binders of the same high-temperature PG grade. Results are shown in figure 12. More binders and mixtures were tested for the comparison between the two parameters, as shown in figure 13. The figure shows that |G*|/sinδ is not a significant controlling parameter. Repeated shear creep and recovery tests focusing on the irrecoverable accumulated deformations were explored, refined, and recommended as an improved protocol over |G*|/sinδ by improving the correlation between the two mix and binder parameters.
1 mm = 0.039 inches
Figure 12. Graph. RSCH data from NCHRP 9-10.(20)
1 Pa = 0.000145 psi
Figure 13. Graph. Modified binder properties and mixture permanent deformation from NCHRP 9-10.(20)
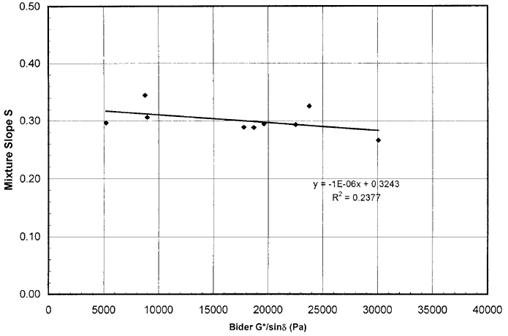
Laboratory fatigue tests on mixtures were also conducted and compared to the SHRP binder specification |G*| × sinδ. As shown in figure 14, little correlation was found. In a similar fashion, cyclic fatigue tests similar to those normally conducted on asphalt mixtures were conducted on binder, refined, and recommended as an improved protocol over |G*| × sinδ. This improved the correlation with mixture fatigue tests.
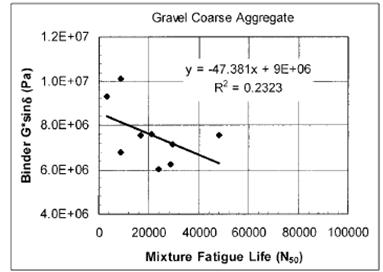
1 Pa = 0.000145 psi
Figure 14. Graph. Modified binder properties and mixture fatigue from NCHRP 9-10.(20)
FHWA conducted a laboratory experiment (internally designated as project 90-07) that used a single crude source and a variety of modification techniques and also confirmed that |G*|/sinδ and |G*| × sinδ do not control mixture permanent deformation and fatigue cracking.
A number of other researchers have also identified shortcomings and recommended alternative protocols. For example, California researchers suggested eliminating the fatigue cracking parameter |G*| × sinδ, increasing mixture characterization efforts, and exploring the binder creep slope parameter used in low-temperature cracking, since fatigue cracking shares similar phenomena identified in original SHRP research.(21)
The extent to which the current Superpave® PG binder specifications characterize the contribution of conventional or unmodified asphalt binders to pavement performance has been shown to depend on the particular dataset at hand. The current system does not effectively characterize the performance of modified asphalt binders. In the interim, State highway agencies have developed a variety of localized specifications added to the Superpave® PG binder specifications that can limit innovation and impact performance.
Proposed changes to the PG binder specifications recommended by NCHRP 9-10 are available.(20) Alternative candidate specification parameters and alternative protocols for performance grading modified and unmodified asphalts are needed. Justification for any changes requires valid evidence from full-scale pavement performance.
TPF-5(019) and SPR-2(174) have been established to study, select, build, and test full- scale accelerated pavement tests to evaluate and validate proposed recommendations to the Superpave® binder specification and to provide AASHTO with a binder purchase specification that is blind to the type of modification. To achieve this objective, different modified materials and configurations were placed in test sections that also investigated other nationally significant and complementary research topics, including the following: