U.S. Department of Transportation
Federal Highway Administration
1200 New Jersey Avenue, SE
Washington, DC 20590
202-366-4000
Federal Highway Administration Research and Technology
Coordinating, Developing, and Delivering Highway Transportation Innovations
| REPORT |
| This report is an archived publication and may contain dated technical, contact, and link information |
|
| Publication Number: FHWA-HRT-12-048 Date: November 2013 |
Publication Number: FHWA-HRT-12-048 Date: November 2013 |
Identifying environmental health and safety considerations associated with the application and removal of pavement marking materials is important to workers, employers, and Government agencies involved with pavement marking. Pavement marking materials contain a variety of chemical compounds and physical characteristics that pose potential risks to human health and the environment during their application, presence in the roadway environment, removal, and disposal. The environmental health and safety risks involved with pavement markings are caused by the marking product chemical composition; equipment used for handling, applying, and removing; and roadway operations associated with application and removal procedures.
This chapter identifies environmental health and safety consideration for handling, applying, removing, and disposing pavement markings. The chapter addresses the following topics:
Pavement marking application and removal techniques have been summarized based on field observations and personal experience of the authors. Material safety data sheets (MSDSs), product-specific factsheets, and/or vendor information provided with products were collected and screened as part of the composition evaluation to formulate a complete list of chemicals and chemical mixtures used in pavement marking products. Environmental health and safety considerations were evaluated based on existing research available in published literature and through the experience of the authors.
Federal environmental and occupational safety regulations pertaining to the pavement markings industry were reviewed to provide regulatory context within the chapter. The following Federal regulations are discussed:
The provided discussion is meant to act as an overview of existing regulatory frameworks that pertain to the industry and not as a comprehensive overview. The process of using LCA to inform decisionmaking is described, and an outline for creating an LCA specific for pavement marking systems is given. In addition to an LCA approach for selecting products, BMPs aimed at reducing environmental health and safety considerations are discussed.
BMPs for reducing exposures during storage, handling, application, and removal of pavement marking products are discussed. Included within the BMP discussion are recommendations for standardizing MSDS reporting practices for pavement marking products and a need for technical specifications for the pavement markings industry. Development of technical specifications for the storage, handling, transfer, application, and removal of marking products will reduce worker exposures and minimize product loss/release to the environment.
PAVEMENT MARKING PRODUCT COMPOSITION, APPLICATION, AND REMOVAL TECHNIQUES
Pavement Marking Composition Overview
Pavement marking materials are either liquid or premanufactured materials that are applied to pavement surfaces to provide pavement markings as defined in "Part 3, Markings" of the MUTCD (23 Code of Federal Regulations (CFR) 655, Subpart F).(3) While the MUTCD calls for pavement markings, the diversity of pavement marking technologies currently in commerce causes difficulty when trying to determine marking products or application processes that offer the highest value in terms of durability and performance.(69,70) Often, the most important consideration in selecting a pavement marking is durability. Durability is defined as how long materials retain their daytime and nighttime visibility. In addition to durability, environmental health and safety issues are important considerations when selecting pavement markings.
The basic components of a pavement marking material are a binder and a reflective element. The binder provides the pavement marking physical presence (day and night) and its color. It also serves as a holder of reflective elements. The binder can be a liquid or a preformed solid that is glued to a surface or melted into a surface. The most important and most prevalent components of a binder are the pigment, resin, and filler. Pigment gives the material color, opacity, and body, as well as the ability to provide retroreflection. A pavement marking typically needs to be white or yellow. How well a pigment accomplishes this task and how it wears is important. However, some of the most effective and durable yellow pigments contain lead and other heavy metals that can create potential environmental and occupational safety issues. The filler is a cost effective measure that supplements the performance gained from the more expensive pigment. Resin is the glue that holds the marking together and gives it durability. For some paints, the resin also allows the markings to be applied in less-than-ideal weather or pavement conditions.
Reflective elements are necessary to improve pavement marking visibility at night. The reflective elements enable the pavement marking to reflect light from a vehicle head light back to the driver. This process is called retroreflection. Retroreflection returns the light back to the light source rather than bouncing the light off the reflective surface and away from the light source. The retroreflective elements are either dropped on a liquid-applied pavement marking (such as paint) or embedded in the material as it is made (such as preformed tape).
Traffic paints are water- or solvent-based paints that are typically sprayed as lines on the surface of pavement. Traffic paints are the oldest and most widely used pavement marking materials in existence. Paint is the most inexpensive of all pavement marking materials, although its cost has increased slightly as new formulations have been introduced and the market has narrowed. Paint is almost exclusively used for long-line applications. The traffic paint market has changed alongside the architectural paint market due to regulatory impacts caused by VOC limits and regulation regarding the use of lead-based pigments.
The primary components of traffic paint are finely ground pigments that are mixed into a resin or binder system. Additives provide additional desired properties. Pigments are mixed with water or solvent in order to apply the paints. Prime pigments within the paint introduce chemical properties such as ultraviolet (UV) stability or physical properties such as color and hiding (the ability of a paint to cover or block out the surface beneath it). Extender pigments or fillers are also commonly used to bring the pigment level up to the required point. Fillers help reduce cost and give the paint consistency, durability, permeability, and scrubability.
Paint pigments and retroreflective elements are held together and to the road surface by a resin. The most common resin in water-based traffic paints are synthetic polymer acrylic-based resins (often referred to as latex paints). Latex paint systems are utilized for their ability to dry quickly following application, which is known as "fast dry." Fast dry is an important characteristic for traffic paint because it allows the traffic paint to be placed on the road with minimal traffic disruption, paint splashing or tracking caused by vehicles, and water washout risk. Paint with a fast drying resin will dry within 1.5 h under a relative humidity of 90 percent; whereas normal resin does not dry under such conditions.
In waterborne paint, water is primarily a diluting agent. It holds the resin emulsion in solution with the other components until the paint has been applied. The drying time may be reduced by adding ammonia or methanol to the paint. Methanol is also an antifreeze and can be added to protect the paint from freezing in its container and storage tanks.
Traffic paint is most commonly applied with a paint spray gun. A conventional spray gun uses air jets in the tip of the paint gun and operates at pressures from 60 to 140 lbf/inch2. Air spray application is commonly called air atomizing. Airless sprayers force the paint through an orifice into the tip of the spray gun at very high pressure (1,500-3,000 lbf/inch2). Airless spraying has become the most common method of applying traffic paint. It has proven to be faster and less troublesome than air atomize spraying, hence its popularity. However, the high pressures associated with airless spraying present additional occupational safety risks. Figure 33 depicts paint application with a truck sprayer.

Figure 33. Photo. Applying paint with a truck sprayer
THERMOPLASTIC PAVEMENT MARKINGS
Thermoplastic pavement marking uses a block and granular material and melts it so that it can be sprayed, gravity extruded, or pressure extruded (often called ribbon extruded) onto pavement as a line. Thermoplastic is a blend of solid ingredients that become liquid when heated and melted. Reflective elements are mixed into the material by the manufacturer and can be applied to the surface following application. Thermoplastic systems become homogenized when heated and agitated. Thermoplastic adheres to hot mix asphalt concrete (HMAC) by forming a thermal bond. When thermoplastic markings are used on PCC, the PCC must first be treated with a liquid-applied primer where the thermoplastic is to be applied. The elements of thermoplastic paints include pigments, reflective elements, fillers, binders, additives, and primer.
Pigment within thermoplastic paints provides color and chemical properties such as UV stability and hiding. Pigments are heat stable, as thermoplastic is often heated to temperatures in excess of 420 °F. Heating does not present a problem for white pigment like titanium dioxide, but it has proven difficult for yellow organic pigments. Yellow pigments containing lead chromate are very effective with respect to heat stability, UV durability, and color. However, organic yellow pigments are less effective than lead chromate yellow pigments in thermoplastic markings. Fillers, such as calcium carbonate, are added to the thermoplastic paints to provide additional volume, improving durability without the higher cost of the additional pigments.
Thermoplastic markings use either hydrocarbon-based polymers or plant- and vegetable-based alkyd (a modified polyester) as a binder. Thermoplastic is usually named for the type of resin used. The hydrocarbon thermoplastics are typically used for long lines along roadways, and alkyd thermoplastics are most often used for short lines (crosswalks, stop lines, legends, and symbols). In order to enhance product application, plasticizers are also added.
Three common methods for applying thermoplastic traffic markings are a spray gun, an extrusion shoe, and a ribbon gun. The spray gun operates much like a traffic paint spray gun. The extrusion process forces the thermoplastic material through a die or shoe riding on the pavement surface. With the ribbon gun, the thermoplastic material is forced through the system, into the gun, and onto the pavement.
Another variation of thermoplastic pavement markings is called profile thermoplastic, which offers improved durability and better visibility in wet pavement conditions. Profiled thermoplastic pavement markings can also be used as a longitudinal rumble strip. Profiled thermoplastic markings are a variation of normal extruded thermoplastic line markings created by providing a bump or an inversion to give the line a "profile" during application.
The profile, if configured at a height of around 0.5 inches, can result in a rumble effect if vehicle wheels come into contact with the line. Profile thermoplastic is often called rumble line. The line, in effect, also becomes a longitudinal rumble strip. Longitudinal rumble strips are used to provide a run-off-the-road crash reduction technique that is in wide use on rural highways in the United States.
The two most common types of profiled thermoplastics are inverted profile markings and raised profile markings. Inverted profile markings are created by rolling a patented rack and pinion wheel over wet (or cooling) thermoplastic. Profiling gives the line a corrugated appearance. Raised profile markings are created by extruding a thermoplastic marking of normal thickness with a raised thermoplastic "bump" at a uniform spacing.
Melt-In-Place Preformed Thermoplastic Tape
Melt-in-place preformed thermoplastic tape is a preassembled thermoplastic laminate, which is placed on the pavement surface and then melted into the surface via a heat source such as a propane torch. Preformed thermoplastic markings are manufactured in shapes ready to use on pavement and are typically used for symbols at intersections or other pavement identification uses. Preformed tapes do not have any preapplied adhesive, and bonding to the pavement is achieved thermally. There are two basic types of preformed thermoplastic markings: one does not require preheating the road surface, while the other does require preheating the road surface prior to application. The materials used in preformed thermoplastic traffic markings are the same as regular thermoplastic, except they have already been combined into a preformed tape that does not require application equipment. Preformed thermoplastic markings are typically shipped with some reflective elements within the laminate material. Additional reflective elements are normally added to the surface of the material during application.
Application of thermoplastic traffic markings requires that the marking be heated once it is placed on the pavement. Applying the marking to HMAC requires heat only. Applying preformed thermoplastic to PCC requires the use of a primer as well as heat on the PCC. Figure 34 shows a thermoplastic application.

Figure 34. Photo. Applying preformed thermoplastic
TWO-COMPONENT PAVEMENT MARKINGS
Two-component pavement markings are pavement marking systems that form a solid when mixed together and sprayed or extruded onto the pavement. The most common examples of two-component pavement marking systems are thermosets (i.e., epoxy, polyester, and modified epoxy), polyurea, and MMA.
Epoxy paints are used as pavement marking materials to increase durability. As a two-component material consisting of a pigmented resin base and a hardener, epoxy paints are cured by an exothermic thermoset chemical reaction. Reflective elements are easily added during application. Epoxy paint pavement markings use pigments to impart color to the marking. The pigments are ground and mixed into the resin material. Color stability under UV exposure is often difficult to achieve with epoxy markings. Because of this characteristic, the pigment loading has to be much higher than with most other types of pavement marking technologies. The high pigment loading increases the cost of epoxy markings in comparison to other markings. The amount of yellow pigment used in epoxy is also often three to four times more than yellow traffic paint or yellow thermoplastic, which is a concern if lead-based yellow pigments are incorporated into the marking.
The hardener is mixed with the epoxy resin in a mixing tube or an impingement chamber. It is then sprayed to form a durable pavement marking. Proper mixing of the two components requires adherence to product-specified volume and temperature requirements. Mixing the two components can be complex and expensive in terms of equipment and materials. Reflective elements are incorporated into the epoxy marking by spraying the elements onto the epoxy as the epoxy is being sprayed onto the roadway. The element application spray gun is located behind the epoxy spray gun on the striping truck. During application, the two components (resin and catalyst) are mixed together prior to installation. The material is then sprayed, with the reflective elements, onto the roadway. A limitation to epoxy paint is that epoxy should not be applied to a wet surface.
Polyurea markings are two-component durable pavement marking materials that are sprayed. Polyurea materials are marketed as fast-curing systems. Some polyurea materials must be applied by a special striping apparatus, while others can be applied by a standard epoxy truck.
Modified urethane is a two-component durable marking material with similar performance characteristics to those of polyurea and epoxy. The product is marketed as being slightly more durable than epoxy but with quicker cure times and better UV color stability. Modified epoxy and urethane can be sprayed from any standard epoxy truck.
MMA is a two-component durable pavement marking material. It is manufactured in two basic mix configurations: (1) an impingement process (forced together by pressure in a mixing tube or chamber) or (2) a static mixer immediately prior to application. MMA can be sprayed or extruded onto pavement. The material forms a strong bond to the pavement surface by an exothermic reaction (release of heat) that occurs during the mixing process and is finished once applied. MMA was originally marketed primarily as an environmentally friendly alternative to solvent-borne paints in areas where lower temperatures are an issue (i.e., Alaska or northern States in the continental United States). However, MMA has been shown to provide a much longer service life than standard traffic paint and is now considered to be as durable as thermoplastics and tapes.
MMA pavement markings are designed to be resistant to oils, antifreeze, and other common chemicals found on roadway surfaces. MMA reportedly bonds well to concrete pavements, and it requires special equipment for application. Figure 35 shows structured MMA application with a hand striper.

Figure 35. Photo. Applying structured MMA
Cold-applied preformed tape is a preassembled laminate that has a pressure-sensitive adhesive. The tape is rolled out and glued to the pavement surface. Some tapes are flat, while others have structure that enhances the durability and wet pavement performance of the tape's retroreflection. Preformed tapes consist of pigments, resins, liquid primer or contact cement, and reflective materials and can be provided with or without adhesive. Preformed tapes are delivered in rolls and are designed for lane lines, legends, symbols, and transverse markings.
There are three types of preformed tape markings: permanent, temporary removable, and temporary non-removable. Permanent pavement marking tapes can be either flat or patterned and may require the use of a primer or sealer as part of the installation (based on vendor instructions). Permanent tapes are generally used for longitudinal edge lines, skip lines, stop lines, crosswalks, legends, and symbols. Two of the most common types of plastics binders used for permanent tapes are urethane and pliant polymer. Temporary tapes are typically used for construction or other short-term traffic management situations.
Pigments used in preformed tape pavement markings impart color to the marking and are ground and mixed into the resin tape during fabrication. The tape is held together with prereacted resins that hold the reflective elements and pigments in place. Some tapes provide an adhesive backing on the bottom side of the resin for adhesion to the roadway surface. Others require that a primer or additional adhesive be applied in the field. Because preformed tapes are manufactured with reflective elements built into the tape, no additional reflective elements are added in the field.
Tape is applied directly on the surface and bonded with an adhesive. Pressure-sensitive adhesives work best when overlaying permanent tapes on new asphalt pavement surfaces. Contact cement or primer is often used when installing the tape on concrete or over older markings. Markings are initially bonded with a light hand roller or vehicle tire and permanently bonded by traffic wear. Figure 36 shows an installed preformed pavement marking tape.

Figure 36. Photo. Marking tape in recessed pavement groove
Glass beads are the most commonly utilized reflective elements used in pavement markings. Glass beads are used to provide improved visibility of traffic markings at night. Beads are embedded into the traffic marking material and reflect light from a vehicle's headlights back to the driver. Spherical reflective beads are retroreflective when embedded in a traffic paint material to a depth of approximately 50-60 percent of their diameter. When light strikes a bead, it is refracted and reflected. The refractive index (R.I.) represents how much a bead bends the light. The bedding for the bead (paint, thermoplastic, etc.) acts as a mirror and allows the light to be reflected. Therefore, the depth of embedment in the marking material has a significant effect on the retroreflective properties of the bead.
Beads used for pavement markings are typically made with an R.I. of 1.50, 1.65, or 1.90. The amount of embedment in the marking material depends on the size of the glass bead, the thickness of the dry marking material, and the application process. Some pavement marking manufacturers use two sets of beads, each with a different R.I. One bead is designed for dry retroreflectivity and the other for retroreflectivity in wet weather. Water on a marking material will change the R.I. of the bead. Therefore, using one bead with an R.I. for dry weather and another with an R.I. for wet weather can result in a marking material that maintains its retroreflective performance in both wet and dry conditions.
Glass beads used as reflective elements in pavement marking systems are commonly grouped into three types following AASHTO M247 and FHWA's Standard Specification for Construction of Roads and Bridges on Federal Highway Projects (FP-03 Section 718-19).(33,71) Type 1 beads are the smallest and are used only as intermix beads for thermoplastic. Type 2 beads are commonly referred to as standard beads because they have historically been the typical drop-on beads used by transportation departments. Type 3 beads (and higher) are drop-on beads of a larger gradation. They are relatively new to the market and are marketed for their ability to provide wet-nighttime visibility under certain conditions. Type 2 beads (and higher) are sometimes coated with a moisture-proof adhesion or floatation coating to help them properly embed in the marking material and avoid clumping together. Type 1 beads are typically not coated when used in an intermix thermoplastic application.
During bead application, it is essential to control the amount and dispersion of beads reaching the marking material and the depth of embedment in the marking. The bead amount, dispersion, and embedment are affected by the bead drop rate, speed of the striping truck, temperature, and viscosity of the binder material. Figure 37 shows a bead gun with a bead shroud dropping beads on ribbon-extruded thermoplastic pavement markings. Figure 38 shows a two-component pavement marking material with a double drop of glass beads applied with standard flare nozzle guns.

Figure 37. Photo. Glass beads applied with bead gun on thermoplastic

Figure 38. Photo. Double bead drop with flare nozzle guns on a liquid material
A typical glass bead application requires significant excess bead applications in order to create the correct bead density in the pavement marking. Bead drop rates usually range from 6 to 12 lb per 100 ft2 for thermoplastics and are often higher for paints and epoxies. Figure 39 shows "clouds" of excess beads created during a bead drop application. Most of the pavement marking glass beads that run off the roadway are associated with application process overspray and excess beads that do not embed in the marking material.

Figure 39. Photo. Glass bead application on a highway on the Tennessee test deck
The chemical and physical properties of glass beads are controlled during the manufacturing process. The most important properties for beads used in pavement markings include bead size, R.I., clarity, and roundness. Factors that affect these properties include the type, quality, and clarity of the virgin or recycled glass used to create the bead; furnace type and temperature; and sieve size. The size of a glass bead affects retroreflective performance, especially under wet conditions, but has no effect on R.I. Instead, the large beads have better performance under wet conditions because their higher profile can protrude through a thin film of water better than small beads. If the layer of water becomes thick enough, large beads will also be ineffective.
Glass bead retroreflective performance is also related to the materials used to manufacture the glass. The primary compound for manufacturing glass is silica, but other substances are often added to simplify the manufacturing process and improve the qualities of the produced glass (including carbonates, oxides, industrial by-products, and recycled glass). While beneficial reuse of recycled or by-product materials can save raw materials and energy, the added components may also introduce impurities into the product. Figure 40 shows glass beads embedded in an MMA pavement marking.

Figure 40. Photo. Glass beads in MMA
Proprietary Reflective Elements and Bead Clusters
While glass beads dominate the market for reflective elements in pavement markings, several companies have developed alternatives to glass beads. One company has developed a microcrystalline ceramic bead that serves as a reflective element. Others have developed "bead clusters." Alternatives will likely continue to penetrate the reflective element market in the near future.
PAVEMENT MARKING REMOVAL (ERADICATION)
The most common methods of pavement marking eradication (or removal) include blasting (hydro, sand, or shot), grinding, and masking (using paint, black tape, slurry, or a surface treatment that covers or encapsulates the marking). These eradication techniques can be used individually or in combination. The technique used to remove pavement markings is chosen based on the type of marking to be removed, the pavement surface type, the location of the marking to be removed (as it pertains to the needed path of travel for traffic), and past experiences with eradication techniques.
Removing pavement markings presents unique challenges based on each marking pavement surface scenario. For instance, abrasive blasting or high-speed grinding does not work well on thermoplastic markings on most surfaces because heat produced from the blasting or grinding will sometimes melt the thermoplastic rather than remove it. Grinding is not a particularly good choice for PCC because it removes the texturing of the pavement surface and can lead to major pavement scarring. Preformed marking tape removal is also a challenge when using blasting or grinding, as the tape can break up into small flakes that contain enough adhesive to adhere to the removal equipment or the pavement. Hydro-jetting or hydro-blasting, if used, results in slick pavements in the wintertime, and application of heat can make HMAC slick. When masking is used, the process can sometimes scar the pavement in a fashion that appears to be a traffic marking under certain viewing conditions.
Waste production during eradication techniques also presents a challenge. A significant amount of waste material can be produced when removing markings. Solid debris must be collected from the roadway surface and properly disposed when removing thicker applications of thermoplastic and MMAs. Chemical concerns from stripping agents and some marking products themselves may also be realized when the markings are removed. Marking eradication can also result in significant dust generation, which can impact roadway visibility and can be an occupational safety concern.
While no individual eradication method is free from challenges, the need for pavement marking eradication will continue. Additional research into pavement marking eradication based on pavement marking chemistry or manufactured degradability over time is warranted. New eradication techniques also offer opportunities within the current market.
ENVIRONMENTAL HEALTH AND SAFETY CONSIDERATION CONCERNING PAVEMENT MARKINGS
Environmental health and safety considerations during the application, use, and removal of pavement markings include reducing occupational exposure to the chemical components of the pavement markings and to fugitive emissions of vapors and particulate matter (PM) generated during application and removal of the products. Methods to reduce occupational exposures to the marking products should be taken into account when developing a health and safety program and can easily be accommodated in existing programs that aim to reduce occupational exposures to traffic, which remains a greater acute risk to workers than the products themselves. However, enough evidence exists to support putting into place protective measures that will reduce long-term chronic exposures to marking products.
Occupational exposure to pavement marking chemicals or emissions can occur during the storage, handling, and application of markings; during cleanup of the application devices; and from waste produced throughout the process. Exposures will increase in the event of spilled materials, emergency situations caused by equipment failures or traffic crashes, or poor personal protective equipment (PPE) use.
Potential environmental health and safety concerns include chronic exposures to several chemicals present in pavement marking systems as declared on MSDSs provided by vendors along with the pavement marking products used during the Tennessee and Alaska field demonstration projects. Table 60 presents a summary of chemicals within pavement marking products acquired for the field demonstration project. Appendix E includes a full list of products reviewed.
Table 60. Representative list of chemicals in pavement marking products used in Tennessee and Alaska field demonstration projects based on MSDS review.
Chemicals from MSDS Information |
Hazardous |
Hazard Listing and |
|---|---|---|
Acetone |
Yes |
F-003, 40 CFR 261.31; U-002, |
Acrylated urethane |
No |
Not hazardous |
Alkyl glycidyl ether |
No |
Not hazardous |
Barium sulfate |
No |
Not hazardous |
Bisphenol-A-(epichlorhydrin) epoxy resin |
No |
Not hazardous |
Dibenzoyl peroxide |
No |
Not hazardous |
Dicyclohexyl phthalate |
No |
Not hazardous |
Diethylenetriamine |
No |
Not hazardous |
Diglycidyl ether of bisphenol |
No |
Not hazardous |
1,6-diisocyanatohexane homopolymer |
No |
Not hazardous |
Butyl methacrylate |
No |
Not hazardous |
Hexamethylene diisocyanate |
No |
Not hazardous |
2-ethylhexylacrylate |
No |
Not hazardous |
Limestone |
No |
Not hazardous |
Methanol |
Yes |
F-003, 40 CFR 261.31; U-154, |
MMA |
Yes |
U-162, 40 CFR 261.33 |
4-nonylphenol |
No |
Not hazardous |
Modified polyamine |
No |
Not hazardous |
Polyurethane |
No |
Not hazardous |
Silica (quartz/crystalline) |
No |
Not hazardous |
Titanium dioxide |
No |
Not hazardous |
2,2,4-trimethylpentane-1,3-diol monoisobutyrate |
No |
Not hazardous |
Trimethylolpropane triacrylate |
No |
Not hazardous |
Urethane acrylate |
No |
Not hazardous |
Xylene |
Yes |
F-003, 40 CFR 261.31; U-239, |
The primary concerns revealed through the evaluation of the MSDS include dermal and inhalation exposures of solvents (including acetone, methanol, and xylene), bisphenol-A, nonylphenol, phthalates, and MMA. Long-term exposures to solvents at low to moderate exposure levels is known to cause eye, skin, and respiratory system irritation, headaches, dizziness, and nausea. Long-term exposure to endocrine-disrupting compounds, including bisphenol-A, nonylphenol, and phthalates, is documented for environmental endpoints, but it is less understood for humans. Exposure to MMA causes similar symptoms to exposure to solvents, with additional concerns for impact on the nervous system of an exposed individual. While there is potential risk associated with the materials used in product formulations, the actual level of risk is not characterized.
Additional environmental health and safety concerns have recently been raised due to the heavy metals content in recycled glass beads used as reflective elements. Two existing studies highlight the loss of heavy metals, including arsenic, lead, and antimony, from the surface of the beads under laboratory conditions. The reported concentrations of total arsenic and lead in the beads are significantly high enough to raise concern. The two primary studies concerning the presence of heavy metals in recycled glass beads include the New Jersey Department of Transportation (NJDOT)-funded New Jersey Institute of Technology/Rowan University (NJIT/RU) heavy metals leaching study and the American Glass Bead Manufacturing Association (AGMBA)-funded TTI/ Texas A&M University glass beads leaching study.(72,73) Both the NJDOT and TTI studies observed elevated arsenic concentrations in the glass beads and in leaching solutions mixed with the beads. A summary of both studies is included in this section, along with a summary of a subsequent study performed at FHWA that aimed to contextualize the NJDOT study. An overview of additional research being carried out on this topic at TTI with Texas A&M University, Scientific Applications International Corporation (SAIC), Chalmers Engineering, and the Environmental Protection Agency (EPA) aiming to assess the risk of the metals within the glass beads is also provided.
NJDOT GLASS BEAD STUDY OVERVIEW
Research sponsored by NJDOT and FHWA was conducted by NJIT/RU to study the environmental implications of using glass beads containing elevated concentrations of metals and metalloids on roadways. The study reviewed applicable literature, evaluated laboratory analytical procedures for measuring metals in the glass beads, and reported on leaching of metals from glass beads. The NJDOT study identified the following summarized technical objectives as the basis for the research:
The report introduced AASHTO standard M247-09, Standard Specification for Glass Beads Used in Pavement Markings, and the State-adopted standards in accordance with the first two objectives of the study.(72,33) The report continued with an extended review of the glass bead manufacturing process and metals leaching for reference.
The report evaluated two procedures for measuring metal concentrations in glass beads. It compared the total metal concentrations in the beads measured using findings between bead digestion with hydrofluoric acid followed by measurement with inductively coupled plasma mass spectroscopy versus measurement with a field-portable x ray fluorescence (FP-XRF) detector. The authors determined that the total metals concentrations using both methods were comparable and promoted using FP-XRF because of the method's rapid use and lower safety risk. Results of the bulk bead metal analysis showed a wide range of metal concentrations in the beads tested, ranging from 0.07 parts per million (ppm) for beryllium to 1,120 ppm for barium. Antimony, lead, and arsenic concentrations ranged from 54-192, 19-204, and 92-823 ppm, respectively.
The report also highlights findings from three individual leaching procedures applied to the beads. The procedures included the fractional factorial method, the toxicity characteristic leaching procedure (TCLP), and the synthetic precipitation leaching procedure (SPLP). For the factorial study, the researchers investigated factors affecting leaching, including pH, chemical (salt) application, ionic strength, and time. The tests simulated the type and amount of salt typically used on roadways. The factorial study also simulated weathering and wear and tear of roads by grinding the beads to smaller sizes. TCLP was used to determine whether the beads would be classified as hazardous waste under Federal regulations. SPLP was used to assess the risk of groundwater contamination posed by land application of solid wastes. SPLP extracts were interpreted to represent the leachate potential leaving the applied material and were gauged against criteria for assessing risk to groundwater.
The results of the leaching studies show that 2 to 3 percent of total metal within the glass beads leaches from the beads into the leaching solutions over 160 days. Concentrations within the leaching solutions ranged from non-detectable (nd) to 6,200 micrograms per liter (µg/L) for arsenic, nd to 520 µg/L for lead, and nd to 130 µg/L for antimony (note: 1 g = 0.0353 oz and 1 L = 0.908 quarts). The factorial study results show an effect of pH, ionic strength, and particle size on metals content observed in leaching solutions. Comparison of the leaching procedures revealed that two orders of magnitude lower metals leaching occurs with the 18 h TCLP and SPLP tests than with the 160-day factorial leaching studies.
The study postulates that glass beads may leach metals under field conditions even though significant amounts of metals were not detected in leaching solutions when TCLP or SPLP methods were used. The study also reports that metals concentrations within evaluated batches were very variable. Variability may partly explain why the study did not find a correlation between initial metals concentrations and concentration of the same metals in leaching solutions. Finally, leaching results were compared to the New Jersey groundwater quality standards. Based on statistical analysis and extrapolation of the data, recommended levels of metals within the beads that would result in a leachate metals concentration below the groundwater quality standards were provided.
TTI HEAVY METALS IN GLASS BEADS STUDY
Concerned over the presence of heavy metals in recycled glass bead pavement marking products, AGBMA sponsored research at TTI to determine the composition and leaching potential of heavy metals in recycled glass beads. The TTI study evaluated the presence of arsenic (As), cadmium (Cd), chromium (Cr), copper (Cu), nickel (Ni), and lead (Pb) in three separate batches of type I AASHTO M247 beads and leachate from column studies conducted under laboratory conditions.(33)
The bead total metals content was determined by digesting the beads using Pacific Northwest Laboratory's KOH fusion method and analyzing the resulting solutions for metals according to EPA Method 6020A.(74) Mean ± standard deviation metals contents for the provided samples are shown in table 61 and illustrated in figure 41.
Table 61. Mean ± standard deviation metals contents measured in glass beads (µgmetal/gbead).
Bead |
As |
Cd |
Cr |
Cu |
Pb |
Ni |
ΣÂ Metals |
|---|---|---|---|---|---|---|---|
Batch 1 |
83.3 ±1.42 |
5.92 |
35.8 ±1.32 |
8.88 † |
21.7 |
31.1 ±5.85 |
186 ±3.56 |
Batch 2 |
308 ±23.6 |
nd |
nd |
nd |
77.4 ±27.7 |
76.4 ±4.17 |
462 ±18.3 |
Batch 3 |
393 ±6.53 |
nd |
23.7 |
nd |
81.0 ±7.98 |
nd |
497 ±7.29 |
† = Not all replicates were detectable.
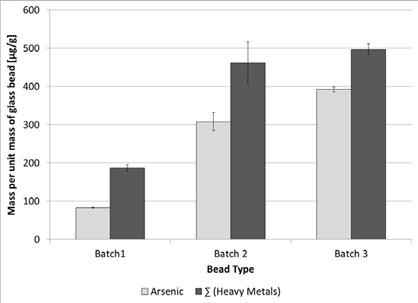
Figure 41. Graph. Mean ± standard deviation arsenic and total metals contents for the samples provided by AGBMA
Arsenic content in the beads accounted for 45 percent of the total measured metals content of batch 1 beads up to 79 percent of the total measured metals content of batch 3 beads. The lowest measured arsenic content (83 µg/g in batch 1 beads) was higher than the metals content of any other metal analyte in all the measured samples. Only nickel in batch 2 and lead in batches 2 and 3 had contents above 50 µg/g. All other metal contents observed for the other analyses were below this level.
Following the composition study, a column leaching system was used to investigate the effect of column eluent solution pH, UV light exposure, temperature exposure, and abrasion on metal release from the beads. The experimental factors and the examined values used in the leaching study are presented in table 62. A complete summary of the experimental study design is available in the TTI project report.(73)
Table 62. Experimental design for the column leaching experiments.
Experimental Factor |
pH |
UV Exposure |
Temperature |
Particle Size |
|---|---|---|---|---|
Evaluated levels |
4 |
12 |
70 (ambient) |
< 149 |
7 |
24 |
100 |
149-250 |
|
10 |
48 |
150 |
> 250 |
|
Bead batch evaluated |
1,2,3 |
1 and 3 |
1 and 3 |
1 and 3 |
1 µm = 0.039 mil.
Results of one-way ANOVA and Tukey's multiple comparison testing indicate that solution pH and abrasion affect the mean amount of arsenic and total metals leached into solution. However, no effect on the mean amount of arsenic or total metal leached was observed due to temperature or UV exposure. The overall amount of total metal released during the 48-h column studies under each experimental condition evaluated was less than 0.2 percent of the total metal content of the beads. However, arsenic concentrations in the column effluent samples were ≥ 100 µg/L for batch 1 and ≥ 3,500 µg/L for batches 2 and 3 during the first hour of collection. After 1 h, observed metal concentrations in the column effluent decreased rapidly. The amount of leaching observed was also greater for smaller particle size fraction samples. Therefore, the fast initial metals release coupled to the finding that smaller particle size fraction releases more metal into solution indicates that leaching is occurred from the surface layer of the bead and not the bead's interior in the experimental setup used in this work.
The TTI study established the presence of heavy metals in recycled glass beads used in pavement marking systems and their potential for leaching. Based on the observed metals content in the beads and in the leaching solutions, additional research was advised. The study recommended that special attention be focused on arsenic due to the magnitude of arsenic content observed in the beads and the high arsenic concentrations observed in the resulting leaching solutions.
The FHWA study established correction factors for heavy metal leaching from glass beads in order to apply laboratory leaching study results to roadside environments. Specifically, the FHWA study was formulated to put the recommendations of the NJDOT laboratory leaching study into a broader context. The study examined the overall size distribution of glass particles in roadside soils and the overall mean percentage of glass as a constituent of the soil.
FHWA obtained roadside soil samples from nine highways, three each in Iowa, Texas, and Virginia. The sampling sites were located adjacent to two-lane two-way highways with edge lines and centerlines marked on an annual basis with paint and glass beads. Three non-overlapping sampling sites 164 to 328 ft long that extended 1.64 to 6.56 ft from the edge of the paved area were established for each highway. Samples of 3.5 oz were collected from 30 10.764-ft2 sampling spots randomly selected from each sampling site. Soil was collected from the top 5.91-inch soil layer and particles greater than 1.57 inches were discarded. The 30 samples collected from each sampling site were mixed together, air dried, sieved through a 0.19-inch sieve, and homogenized in 1.75-oz subsamples using a sample splitter. The glass particles were separated from the homogenized samples and weighed to determine the overall mean percentage of glass as a constituent of the soil. The particle size distribution of the glass beads in the homogenized samples was determined using a series of sieves. The correction factor was calculated based on the overall distribution of glass particle size in roadside soils and the overall mean percentage of glass as a constituent of the soil.
Based on the results of the roadside evaluations in Texas, FHWA proposed a correction factor of 0.002. Multiplying the NJDOT leaching study results by the FHWA correction factor resulted in an arsenic leachate concentration that was more than 20 times below the allowable criterion (in this case, the criteria for ground water). The leachate lead concentration was lower than the allowable criterion for lead in groundwater before applying the correction factor. The FHWA study also recommended that leaching procedures be conducted on roadside soil and not on individual components of the soil (in this case the glass beads) as glass beads comprise less than 0.3 percent of roadside soil.
In response to an FHWA request, TTI proposed a study aimed at supporting decisionmaking concerned with regulating the presence of heavy metals (specifically arsenic and lead) in recycled glass beads used in pavement marking systems. The study is funded by FHWA but has oversight and input from EPA. The study is now being carried out at TTI and Texas A&M University with support from EPA, SAIC, and Chalmers Engineering Services Company. The first objective of the study was to develop a conceptual risk assessment model identifying exposure scenarios likely to occur under occupational and residential conditions. A parameters list formulated from existing exposure assessment models will be refined through field observations related to bead storage, handling, and application procedures on the roadway environment in order to develop a conceptual model. A screening level risk assessment based on the conceptual model of exposure, available literature data, and some base assumptions will be performed utilizing SAIC software and databases.
The second objective of the study examines the relationships between total, extractable, and bioavailable metals content in samples of glass beads. Fifteen batches of AASHTO M247 type I glass beads were under evaluation.(33) Heavy metals in the glass beads were extracted using three separate and independent methods. The total metals fraction in the beads was extracted according to the Pacific Northwest Laboratory's KOH fusion method. The extractable fraction was extracted according to EPA Method 3050B.(75) The bioavailable fraction was extracted with a solution of 0.4 M glycene adjusted to pH 1.5 with hydrochloric acid. The extracts will be analyzed for arsenic and lead following EPA Method 6020A.(74) The results will be compared using corollary statistics (e.g., Pearson's product-moment correlation coefficient) and inferential statistics (e.g., ANOVA followed by multiple comparison testing) to evaluate the likelihood of predicting the bioavailable content in glass beads through analysis of total or extractable metals.
The final objective of the study was to investigate whether the retroreflectivity of the beads was correlated to the metal content of the bead. TTI created pavement marking draw-downs (18-inch pavement marking samples created in the lab under controlled conditions) for the evaluation. The draw-downs are identical except for the beads used. The beads were the same 15 batches of type I beads used to accomplish the second objective as previously described. Three replicate draw-downs were produced. The retroreflective performance of the draw-downs were measured at the standard 98-ft geometry.
The combined results of the three objectives will inform the decisionmaking process regarding maximum allowable concentration of heavy metal in recycled glass beads intended for use in pavement marking systems.
HEAVY EQUIPMENT AND TRAFFIC HAZARDS
While considerations should be taken into account for reducing handling and exposure to pavement marking products, it is important to note that the environment for workers involved in pavement marking poses significant hazards. In addition to possible chemical exposure, installation and removal of pavement markings requires the use of heavy equipment and heating devices.
Traffic hazards also pose a significant hazard when working with traffic markings. By nature, traffic markings are used on traveled roadways. Work zone hazards on roadways are well documented. Even with lane closures and speed limit reductions, workers are often close to high-speed traffic when installing or removing traffic markings. Heavy equipment and traffic hazards are as much of a concern to worker safety as is chemical exposure.
FEDERAL ENVIRONMENTAL AND SAFETY REGULATIONS PERTAINING TO PAVEMENT MARKINGS
Due to potential health and safety considerations posed by the storage, handling, and use of pavement marking products, a review of existing Federal and State environmental laws and regulations is included in this chapter. The information is not meant to be an comprehensive review of all Federal acts, regulations, or policies that may pertain to glass beads. Rather, it is meant to provide background material to put potential proposed regulations into context. This section reviews primary environmental acts as they pertain to pavement markings, including NEPA, TSCA, RCRA, CAA, the Clean Water Act. This section also provides information concerning the Occupational Safety and Health Act and the Federal Hazardous Materials Transportation Law.
NEPA became a law in 1970. Among its provisions is the requirement for environmental reviews of all major Federal actions and decisions. Regulation 23 CFR 771, Environmental Impact and Related Procedures, addresses FHWA actions under NEPA.(76) Section 23 CFR 771.101, "Purpose," states the following:(76)
"This regulation prescribes the policies and procedures of the FHWA and the Federal Transit Administration (FTA) for implementing the National Environmental Policy Act of 1969 as amended (NEPA), and supplements the NEPA regulation of the Council on Environmental Quality (CEQ), 40 CFR parts 1500 through 1508 (CEQ regulation). Together these regulations set forth all FHWA, FTA, and DOT requirements under NEPA for the processing of highway and public transportation projects."
Section 23 CFR 771.117 sets forth the FHWA categorical exclusions under NEPA, and pavement marking projects are typically given an exclusion from environmental review. Subsection 23 CFR 771.117 (c)(8) provides a categorical exclusion for the "Installation of fencing, signs, pavement markings, small passenger shelters, traffic signals, and railroad warning devices where no substantial land acquisition or traffic disruption will occur."(76) In most circumstances, pavement markings would not trigger an environmental assessment under NEPA. However, the applicability of NEPA to a project must be considered for each project, and pavement markings may be involved in an environmental assessment if some other aspect of the project triggered an environmental assessment requirement.
NEPA requirements come into play when Federal agencies are involved in funding, permitting, licensing, or making decisions that can affect the environment. The primary tools under NEPA are the environmental assessment and environmental impact statements, which include processes designed to assess the likelihood of impacts from alternative courses of action.
TSCA provides a mechanism for EPA to identify, list, and categorize new and existing chemicals used in manufacturing and commerce. The primary purpose of TSCA is to identify potentially dangerous products or product uses that should be subject to Federal control. Because of their inorganic and organic chemical composition, pavement marking products are required to meet compliance with inventory listing requirements established in TSCA.
RCRA provides EPA with the authority to control the generation, transportation, treatment, storage, and disposal of hazardous waste. Under the current regulatory framework within the act, it is unclear whether or not pavement marking products or waste from pavement marking products would be considered hazardous wastes. Table 96 through table 103 in appendix F list chemicals contained in pavement marking materials, including chemicals that also are considered hazardous waste upon disposal such as acetone, methanol, MMA, and xylene. In addition to these solvents, the reported levels of lead and arsenic observed in glass beads used as pavement marking reflective materials are characteristic of a hazardous waste under 40 CFR 261.21-24.(77) Cleaning application devices or removing pavement marking materials also creates waste products with potentially hazardous properties. In order to determine whether or not a waste is hazardous, EPA calls on subjecting the waste to TCLP (as defined in 40 CFR 261.24).(77)
The Federal CAA affects pavement marking use, application, and eradication due to the potential release of PM, lead, and VOCs to the air. PM and lead are directly regulated under the National Ambient Air Quality Standards (NAAQS), while VOCs are indirectly regulated because of their role in the formation of ozone (a criteria air pollutant along with PM and lead). Under CAA, as amended in 1990, each State must develop a plan describing how it will attain and maintain NAAQS. This plan is called the State Implementation Plan (SIP) and is required under section 110 of the CAA (40 CFR 51, subparts F and G).(78)
In general, SIP is a collection of programs (monitoring, modeling, emission inventories, control strategies, etc.) and documents (policies and rules) that States use to attain and maintain NAAQS. States must engage the public in approving their plans prior to sending them to EPA for approval. Instating engineering controls to limit the release of PM, lead, and VOCs from pavement marking products could be considered within SIPs. For instance, the use of blasting or other removal procedures that produce PM may be restricted in SIP non-attainment areas. Also, the 2008 changes to the lead air quality standard may affect the pavement markings industry if detectable quantities of lead are identified in air emissions from either applying or removing the markings.
Under CAA, the Federal government also gave EPA the ability to identify a list of hazardous air pollutants not currently listed as criteria pollutants. Several components of pavement markings, including arsenic and VOCs, are found on the hazardous air pollutants list. The list is currently regulated under the National Emission Standards for Hazardous Air Pollutants (NESHAP), which requires air pollution sources to utilize maximum achievable control technologies to limit emissions. Within NESHAP, EPA regulates activities such as paint stripping. However, pavement marking removal activities are not covered under the NESHAP's regulations, likely due to the mobile and low-frequency nature of pavement marking removal operations.(79)
VOC emission from pavement markings was found to be minimal compared to other sources. A report published by EPA in 1989 examined the VOC emissions from various types of pavement markings, and the results of the report are included in table 63.(80)
Table 63. Comparison of estimated VOC emissions.(80)
Marking Materials |
Emissions (lb/lane mi-year) Estimated VOC* |
|---|---|
Solvent-based (non-aerosol) |
69 |
Water-based (non-aerosol) |
13 |
Thermoplastic |
Negligible |
Field-reacted polyester |
Negligible |
Field-reacted epoxy |
0.25 |
Preformed tapes without adhesive primer |
0 |
Preformed tapes with adhesive primer |
58 |
Permanent markers |
0 |
*Lane mile refers to a 4-inch-wide solid stripe that is 1 mi long. The average VOC content for water-based paints is 0.76 lb/gal.
Pavement markings are considered non-point sources under the Clean Water Act. The Nonpoint Source Management Program with the CAA provides grant money for States, territories, and Indian tribes to support a variety of activities to control nonpoint sources of water pollution. These activities may include technical assistance, financial assistance, education, training, technology transfer, demonstration projects, and monitoring.
Because storm water management from roadway surfaces is achieved on a local project level and because transportation departments are actively involved in construction projects, many State transportation departments have developed guidance to control point and nonpoint source water pollution associated with construction and maintenance activities. Examples of State programs include the following:
HAZARDOUS MATERIALS TRANSPORTATION
The U.S. Department of Transportation has the authority to control the transportation of hazardous materials through the Hazardous Materials Transportation Law. Regulations promulgated from this law potentially affect manufacturers and striping crews transporting pavement marking materials if the products are considered hazardous. Regulation 49 CFR 171-180 lists the hazardous materials covered within the Hazardous Materials Transportation Law, including identification of a compounds hazard class and shipping information requirements.(91) Some of the chemicals found in the pavement marking products used on the Tennessee and Alaska test decks include acetone, butyl methacrylate, epichlorhydrin, diethylenetriamine, hexamethylene diisocyanate, methanol, MMA, modified polyamine, and xylene. The law also requires incident reporting and emergency response during spills of hazardous materials during transportation. In addition, under 49 CFR 172.504, "General Placarding Requirements," vehicles used for transporting and applying pavement marking materials are subject to Federal vehicle placarding requirements.(91)
OCCUPATIONAL SAFETY AND HEALTH ACT
The Occupational Safety and Health Act founded the Occupational Safety and Health Administration (OSHA) with the goal of protecting the health and safety of workers employed in private and public sectors. Under 29 CFR 1910, OSHA identifies information that must be available to workers and the public regarding the chemical constituents of products.(92) Regulation 29 CFR 1910.1200(g)(1) requires that "chemical manufacturers and importers shall obtain or develop a material safety data sheet for each hazardous chemical they produce or import."(92) This information is typically provided in the format of an MSDS. The format for MSDS reporting has evolved over time and most companies follow American National Standards Institute (ANSI) standard Z400.1, Hazardous Industrial Chemicals-Material Safety Data Sheets-Preparation, which is a voluntary consensus standard for the preparation of MSDSs.(93) Pavement marking products are supplied with an MSDS, and employees working with the products need to be aware of the MSDS and any health and safety considerations of working with the products that are identified.
In addition to requiring MSDSs, OHSA regulations pertain to reducing exposures to particular components commonly found in pavement markings, including lead, hexavalent chromium, silica, and respirable dust. Worker exposure to lead is regulated under 29 CFR 1926.62, and subsection 29 CFR 1926.62(c)(1) establishes a permissible exposure limit (PEL) for lead at a maximum of 50 µg/m3 of air averaged over an 8-h period.(94) OSHA regulation 29 CFR 1910.1026 regulates worker exposure to chromium (VI) and establishes a PEL not to exceed 0.5 µg/m3 as an 8-h time-weighted average under any expected conditions of use.(92)
OSHA regulation 29 CFR 1926.55 also establishes threshold limit values for airborne contaminants for construction activities, including limit values for seven silica compounds and six silicates.(95) Because workers handling, applying, and eradicating pavement marking may be exposed to lead, chromium (VI), or silica, monitoring and worker protection measures should be considered.
In addition to chemical exposures, OSHA also sets requirements for noise protection. Applying and removing traffic markings may require noise protection for exposed workers. The OSHA requirements for noise control and hearing conservation are set forth in 29 CFR 1910.95, "Occupational Noise Exposure."(92) This regulation establishes permissible noise exposures for short time periods (15 min or less) up to a full 8-h day. Noise levels that exceed the permissible noise exposure levels require hearing protection, noise reduction, and, in certain instances, hearing conservation programs.
ENVIRONMENTAL LCA OF PAVEMENT MARKINGS
The choice between two pavement marking products should be made based on the performance of the marking materials and the chemical constituents (determined through reporting requirements, including MSDS). However, if the two products have similar performance characteristics and chemical composition, a review of the product life cycle may be beneficial to reducing environmental health and safety considerations indirectly related to the pavement markings industry prior to making decisions concerning product selection. LCA offers decisionmakers with tools and information to gain a higher level of understanding of the environmental trade-offs associated with a product or process by following a product through production, use, disposal, or reuse. Examples of factors considered during LCA include energy use and consumption, water use, greenhouse gas production, waste and wastewater production, hazardous pollutant generation, toxicity endpoints, and carbon footprinting.
The Organization for Standardization (ISO) provides a framework for LCA through two guidance documents: ISO 14040:2006, Environmental Management-Life Cycle Assessment-Principles and Framework, and ISO 14044:2006, Environmental Management-Life-Cycle Assessment-Requirements and Guidelines.(96,97) Collectively, these two documents describe the principles and framework for conducting LCAs. While the ISO standards provide broad guidance on LCAs, other groups have taken guidance on LCAs significantly further. Table 64 provides existing additional guidance used for LCAs along with the program country of origin.
Table 64. LCA methodologies.(98)
Methodology |
Developer |
Country of Origin |
|---|---|---|
ILCD |
European Commission Joint Resource Centre |
European Union |
CML2002 |
Centre of Environmental Science at Leiden University (CML) |
Netherlands |
Eco-indicator 99 |
PRé |
Netherlands |
EDIP97 - EDIP2003 |
DTU |
Denmark |
EPS 2000 |
IVL |
Sweden |
Impact 2002+ |
EPFL |
Switzerland |
LIME |
AIST |
Japan |
LUCAS |
CIRAIG |
Canada |
ReCiPe |
RUN + PRé + CML + RIVM |
Netherlands |
Swiss Ecoscarcity 07 |
E2+ ESU-services |
Switzerland |
TRACI |
EPA |
United States |
MEEuP |
VhK |
Netherlands |
Note: Reference information for each methodology is provided in ILCD Handbook, International Reference Life Cycle Data System. (See reference 99.) |
||
The first step in an LCA for pavement marking products is to define the goal. A typical goal for an LCA is to compare two materials and determine which has the least overall impact on the environment. For pavement markings, the impact on the environment will arise from extraction of the raw materials used to make the product (commonly including petroleum products as well as inorganics). Transforming the extracted or recycled raw materials into pavement marking products requires energy and water. The production process results in the formation of waste streams that could escape in wastewater and air. Additional impacts are associated with packaging and distributing pavement marking materials and applying pavement marking materials to roadways. Waste products also include not only the original manufacturing materials but also road surface and equipment cleaning wastes.
Potential impacts become part of a life-cycle inventory (LCI). LCI is a process of quantifying energy and raw material requirements, atmospheric emissions, waterborne emissions, solid wastes, and other releases for the entire life cycle of a product, process, or activity. In the LCI phase of an LCA, data are collected and organized. The collected data are necessary to evaluate comparative environmental impacts or potential improvements. LCI documents the quantities of pollutants released to the environment and the amount of energy and material consumed. The four steps in an LCI process are as follows:
Figure 42 shows a schematic of a pavement marking LCA model.
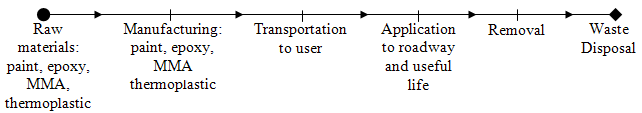
Figure 42. Illustration. Pavement marking LCA model
The life-cycle inventory assessment (LCIA) phase of an LCA is the evaluation of potential human health and environmental impacts of the environmental resources and releases identified during the LCI. An LCIA does not necessarily attempt to quantify any specific actual impacts associated with a product, process, or activity. Instead, it seeks to establish a linkage between a system and potential impacts. LCIA models are based on the models within each of the impact categories using assumptions. The models provide an overview of the relative risk and are not intended to provide specific predictions of risk.
After the LCI and LCIA are completed, the results are subjected to a life cycle interpretation. ISO 14040:2006 defines life cycle interpretation as follows:(96)
"Interpretation is the phase of LCA in which the findings from the inventory analysis and the impact assessment are considered together or, in the case of LCI studies, the findings of the inventory analysis only. The interpretation phase should deliver results that are consistent with the defined goal and scope and which reach conclusions, explain limitations, and provide recommendations."
LCIA models are typically simplified versions of more comprehensive models used in the impact category analyses. These simplified models are suitable for relative comparisons of the potential to cause human or environmental damage but are not indicators of absolute risk or actual damage to human health or the environment. Finally, the interpretation step considers findings from the inventory analysis and the impact assessment together. The interpretation should reflect the relative approach used for LCIA, indicating potential environmental effects rather than predicting actual impacts on category endpoints. The interpretation phase may also be an iterative process of reviewing and revising the scope of the LCA as well as the nature and quality of the data collected in a way that is consistent with the defined goal.
An accurate LCA for a pavement marking material requires detailed information from the manufacturer. A list of ingredients in the marking material and their concentrations is a starting point. The processes used to produce these ingredients must be determined, and their environmental impacts should be factored into the assessment. For example, energy and waste material associated with producing a resin in a marking material becomes part of the analysis. The energy, emissions, and waste products associated with producing the marking material from the ingredients is another step. Transporting the marking materials to a user also requires energy, typically petroleum fuel products, and generates additional emissions. Applying the pavement marking material usually involves some waste material, emissions, and energy (for vehicles or heating requirements). Finally, removing the marking material also requires energy and produces additional emissions and waste products. Quantifying and calculating these elements are the essential tasks in developing a pavement marking environmental LCA.
As of the writing of this report, LCIs for pavement marking products do not exist. However, their development is a potential future focus of research in academics, government, and industry. Developing LCAs for pavement markings is an important step forward and will become a necessary tool to make sound decisions regarding product selection for pavement marking installations.
When the total environmental impact of a pavement marking product is unknown but sufficient concern exists to warrant minimizing exposure to products, implementation of BMPs will help reduce potential adverse outcomes. Occupational exposure to traffic conditions, heavy metals and silica through dermal contact, accidental injection, and inhalation of product dust is potentially significant. If there are concerns that the exposure limits for heavy metals or silica may be exceeded, OSHA procedures for sampling and monitoring the potential exposure should be followed. Additionally, BMPs may be implemented to reduce worker exposure to pavement markings. BMPs for pavement markings are associated with the storage, handling, application, and removal of pavement markings.
Proper storage, handling, application, and removal of pavement markings will minimize the possible release of pavement marking contaminants to the environment and include preventative measures for managing accidental spills of pavement marking materials.
BMPs for minimizing environmental exposures to pavement marking products that can be adopted include the following:
In addition to the BMPs identified to reduce environmental exposures to workers from pavement marking products, reducing hazards associated with the use of heavy equipment and traffic hazards is also important. Hazards associated with pavement marking application from equipment and traffic are similar to hazards faced within roadway construction or maintenance operations. Considerations specific to traffic marking application or removal include worker exposure to roadway environments. Some of the equipment is operated at very high temperatures (above 250 °F) and pressures.
A need for standardizing and improving the information listed on product MSDSs became apparent based on the review of 41 pavement marking product MSDSs (listed in appendix E and summarized in appendix F). Products reviewed were shipped with either ANSI- or OSHA-formatted MSDSs. Within each format, there were significant differences in the information provided. Therefore, developing improved standardization of MSDS information for pavement marking materials will facilitate comparison of the materials and assist with managing environmental risks and impacts.
At a minimum, the following recommended changes should be adopted:
Training pavement marking work crews and managers to read and understand the provided MSDSs is also necessary. Marking crews know that MSDSs are available and are carried along with the team; however, the value of the MSDS in regards to reducing environmental health and safety is not appreciated. There is also confusion regarding what to actually do about the information listed on the MSDS. Training of PPE and BMPs used to reduce exposure to pavement marking products would benefit the industry. These practices should be specified in technical specifications on pavement marking storage, handling, application, and removal that are set through AASHTO or ASTM.
Table 65. MSDS ANSI format.(93)
Section 1. Chemical Product and Company Identification: Names the material and links MSDS to the label and shipping documents. Includes the name, mailing address, and telephone number for the manufacturer or distributor. |
Section 2. Hazards Identification: Descriptions of the material's appearance, odor, and health, as well as physical and environmental hazards that may be of concern for emergency response personnel. Includes four subsections: emergency overview, OSHA regulatory status, potential health effects, and potential environmental effects. |
Section 3. Composition Information on Ingredients: Identifies the hazardous components of the material. Non-hazardous ingredients are listed separately. Chemical Abstracts Service (CAS) numbers are included, as well as percentages or ranges of percentages. |
Section 4. First Aid Measures: Includes two subsections, emergency and first aid procedures, and a note to physicians regarding additional information on antidotes, specific treatments, and diagnostic procedures intended for use by healthcare professionals. |
Section 5. Fire-Fighting Measures: Describes fire and explosive properties of the material, extinguishing media to be used, and fire-fighting instructions. |
Section 6. Accidental Release Measures: Information regarding preventing or responding to spills, leaks, or releases. Information on personal protective equipment, containment equipment, cleanup equipment and techniques, environmental precautions, and specific reporting requirements may be included. |
Section 7. Handling and Storage: Guidelines on safe handling and storage practices. |
Section 8. Exposure Controls, Personal Protection: Discusses exposure guidelines, such as the OSHA PELs, American Conference of Governmental Industrial Hygienist's Threshold Limit Values, and Biological Exposure Indices. Engineering controls and personal protective equipment are also covered in this section. |
Section 9. Physical and Chemical Properties: The material's physical and chemical properties. Properties typically include appearance, odor, odor threshold, physical state, pH, melting/freezing point, initial boiling point and boiling range, flash point, evaporation rate, flammability (solid, gas), upper/ |
Section 10. Stability and Reactivity: Describes hazards associated with stability and reactivity of |
Section 11. Toxicological Information: Presents toxicological information such as acute dose |
Section 12. Ecological Information: Provides information regarding potential environment impacts associated with releasing the material to the environment or in evaluating waste treatment practices. |
Section 13. Disposal Considerations: Recommends disposal methods, including recycling or reclamation. |
Section 14. Transport Information: Classifies information and special precautionary information for shipping the material. Includes U.S. Department of Transportation classifications or an indication that transporting the material is not regulated. |
Section 15. Regulatory Information: Provides information regarding the regulatory status of the material. Addresses regulations under OSHA, EPA, and other relevant regulatory agencies, including State agencies, if appropriate. |
Section 16. Other Information: Lists other material that may be useful and not covered in the preceding 15 sections. May include label information, hazard ratings, revision dates, and references |
SPECIFICATION GUIDELINES FOR STORAGE, HANDLING, APPLICAITON, AND REMOVAL PRACTICES,
The creation of technical specifications for work practices concerning pavement marking products is recommended to address environmental health and safety considerations. Currently, tremendous variation within the pavement markings industry exists regarding how products are stored in contractor, city, or State yards. Product spillage is a concern due to the content and cost of pavement marking products. Standard specifications would allow for better control of pavement marking products in storage and result in fewer spills.
The process for handling product materials and transferring product materials from storage to the application devices is also variable. The technical specifications would be dependent on the type of application (long-line versus short-line applications) and the type of marking being applied. PPE measures could be specified within a guidance document to standardize measures put in place to reduce exposures to products. Similarly, the actual methods of application should be specified to achieve an industry standard of quality across crews applying markings. Currently, quality assurance is a mostly qualitative measure, and the resulting quality of applied markings is highly variable. However, technical specifications could be followed to help standardize the applied marking quality. The specifications, based on the best practices currently available in the industry, would result in fewer environmental health and safety concerns and less product loss/wastage.
Finally, the process for removing marking products must be specified. Currently, State guidance and regulations regarding removal methods is variable. Because the amount of exposure to the products is based on the type of removal occurring, technical specifications requiring vacuum recovery and identifying the appropriate disposal method for the removed materials will be valuable to the industry.
This chapter identifies and highlights important environmental health and safety considerations for the application and use of pavement marking products. Pavement marking product compositions, application techniques, and removal procedures are introduced. Worker health and environmental considerations for paints, thermoplastics, epoxies, tapes, and reflective elements are discussed. Included within the discussion is a summary of existing research seeking to understand the impact of heavy metals present in glass beads used to impart retroreflectivity of pavement markings. The chapter also provides an overview of existing Federal environmental and safety regulations pertaining to pavement markings.
LCA is introduced in order to suggest a framework that can be used to include environmental health and safety considerations into the process of selecting pavement marking products for application. BMPs for storage, handling, application, and removal of pavement marking materials are also introduced in this chapter. The chapter ends with recommendations to standardize the information present on the MSDS accompanying the products and develop specification guidelines for the storage, handling, application, and removal of pavement markings. The appendices provide additional resources, including a summary of the products used within the pavement marking demonstration project.